The in vitro method uses in vitro detection methods to evaluate the potential sensitization of food allergens, which is safer and more feasible than in vivo experimental detection. In current research and practice, in vitro detection methods mainly evaluate the potential sensitization of food allergens from the perspective of serology and cytology.
Food allergen sensitization in vitro experiments include allergen adsorption experiment, western blotting, enzyme-linked immunosorbent assay (ELISA), histamine release experiment and simulated gastrointestinal digestion.
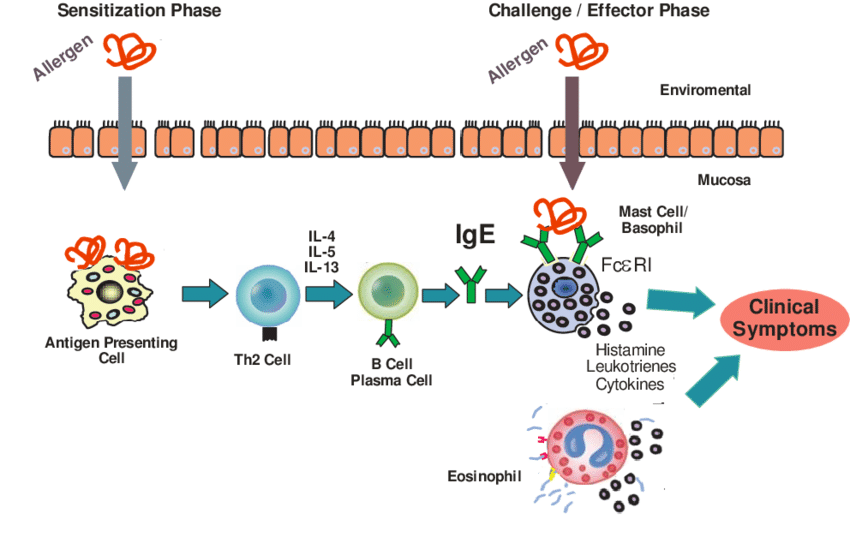
Serological Methods
The serum of allergic patients contains IgE antibodies that can specifically bind to the allergen. The potential sensitization of the protein can be evaluated based on the binding ability of the tested protein to IgE in human serum. In order to overcome the dependence on human serum, some immunoassay methods that rely on the antiserum of animals such as mice and rabbits have gradually developed. Currently commonly used serological methods mainly include radioallergen adsorption inhibition test (RAST inhibition test), enzyme-labeled allergen adsorption inhibition test (EAST inhibition test), western blotting and enzyme-linked immunosorbent assay (ELISA).
RAST and EAST
Inhibition experiments RAST and EAST are currently widely used methods for studying allergic reactions in the world, with high sensitivity and accuracy, and at the same time solve the problem of cross-reactions between different food allergens. Therefore, they are the key technology to evaluate the total sensitization of allergens. However, because the RAST and EAST inhibition experiments rely heavily on human serum, and the serum is difficult to ensure consistency, it is difficult to standardize RAST and EAST; and the radioactive hazards during the RAST experiment are serious and damage the health of the staff. Therefore, in the method for detecting food allergens recommended by FAO/WHO revised in 2001, the above two methods are not recommended. At present, food allergen detection basically uses the UniCAP system for RAST inhibition experiments, which can also perform fluorescence enzyme-linked immunoassay.
Western Blot
Western blotting is a hybridization technique that combines high-resolution gel electrophoresis and immunochemical analysis techniques. This method has the advantages of large analysis capacity, high sensitivity, and strong specificity. However, the operation is complicated and time-consuming, and is often combined with ELISA. Used to detect the allergenicity of allergic foods.
Enzyme-linked Immunosorbent Assay (ELISA)
ELISA is a new technology developed in the late 1990s. The method has high sensitivity, good specificity and easy operation. However, ELISA can only determine the specific binding of antigen and antibody, and cannot determine the sensitization of food allergens, and further testing with other methods is needed to determine the sensitization of food allergens. According to different operations, ELISA is mainly divided into double antibody sandwich method, indirect method, capture method and competition method. Among them, double antibody sandwich ELISA and competitive ELISA are the most widely used. ELISA can not only perform qualitative analysis, but also quantitative analysis. In recent years, ELISA technology has developed rapidly, and its sensitivity and specificity have been improved. Competitive ELISA can also be used to quantitatively detect food allergens. In the EL I S A reaction, the smaller the I C 50 value, the stronger the sensitization of food allergens, and vice versa. When detecting multiple food allergens at the same time, cross-reactions may occur and affect the accuracy of the results. However, sandwich ELISA can overcome cross-reactions among food allergens.
Cytological Methods
In a food allergic reaction, immune cells will undergo a series of changes, including the release of cytokines and T cell proliferation. The sensitization of food allergens can be evaluated by detecting a series of changes in immune cells. Generally speaking, the more cytokines are released, the proliferation of T cells is violent, and the sensitization of food allergens is stronger, and vice versa.
Histamine Release Test (HRT)
IgE-mediated allergic reactions can cause a large amount of histamine to be released in the body, and the amount of histamine released can be measured by fluorescence RAST. However, because the histamine determination must be completed within one day after the blood draw, and the serum source of allergic patients is insufficient, and the experiment is easily affected by the environment and operating conditions, the practical application of this method is greatly restricted.
Basophil Activity Test and Degranulation Experiment
The activity of basophils and the amount of degranulation are closely related to the sensitization of allergens. The stronger the sensitization of the allergen, the stronger the activity of basophils and the greater the amount of degranulation. CD63 and CD203c are often used as markers for cross-linking of allergens and IgE to trigger basophil activation. Studies have shown that the detection of CD63 and CD203c through the kit can effectively diagnose children’s egg allergy and milk allergy. The use of basophil activity test technology to detect food allergens has good specificity and high sensitivity, and the test results for patients receiving antihistamine therapy are still reliable. It is a supplement to traditional quantitative determination of IgE levels and skin experiments. The current challenge is how to standardize this method to make it the mainstream method for detecting the sensitization of food allergens and apply it to the evaluation of the sensitization of food allergens.
T Cell Response Analysis
An important stage of IgE-mediated food allergy is T cell proliferation and the production of cytokines (IL-10, IL-13, IFN-γ and TNF-α). The peripheral blood mononuclear cells of allergy patients contain a small amount of specific T cells, which can be used as a source of T cell clones. By measuring the proliferation of T cells and the release of cytokines, the potential sensitization of proteins and the epitopes of T cells can be studied. Studies have found that when T cells are stimulated by peanut extract, they will produce Th1 and Th2 cytokines, but when they are stimulated again by peanut allergen, allergic individuals will only strengthen Th2 response. Therefore, the response of T cells can be used to evaluate the sensitization of food allergens. This method can be used in conjunction with animal experiments to explain the structure that determines protein sensitization, but T cell response analysis is difficult and time-consuming, and it is rarely used in research.
