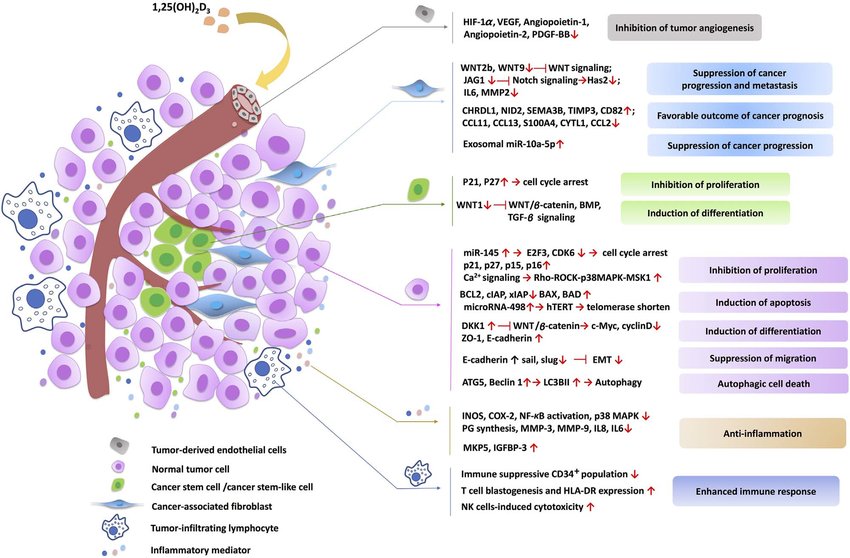
Vitamin D is a fat-soluble vitamin that can be obtained from food. At the same time, cholesterol in the body can be oxidized to 7-deoxycholesterol, which is converted into vitamin D by ultraviolet radiation. It is the precursor of a powerful steroid hormone. It synthesizes biologically active calcitriol after metabolism in the liver and kidneys. Calcitriol binds to the vitamin D receptor (VDR), which can play a variety of biological functions. As an analog of vitamin D, calcitriol has a fast clearance rate in the serum and has little effect on calcium homeostasis. It is widely used in the regulation of calcium and phosphate metabolism. In the relevant research in the past few decades, there is more and more evidence that low sun exposure and vitamin D deficiency are related to the increased risk of cancer. About 80 years ago, it was first discovered that sunlight exposure was negatively correlated with the incidence and mortality of cancer in North America. Later, in 1980 and 1992, epidemiological research reports respectively linked low sun exposure to high risks of colon cancer and prostate cancer for the first time, indicating that vitamin D as a substitute for sun exposure can prevent colon cancer and prostate cancer. Since then, hypotheses about the relevance of vitamin D and cancer have been proposed. Some population studies have shown that serum 25-hydroxyvitamin D (25-OH-D) levels are negatively correlated with the prevalence of colon cancer, breast cancer, prostate cancer, and gastric cancer. The idea that vitamin D deficiency contributes to the occurrence and progression of tumors has become increasingly obvious. Therefore, maintaining a sufficient concentration of vitamin D in the serum may be beneficial to the prevention and treatment of cancer. As more and more epidemiological and clinical studies have observed the beneficial effects of vitamin D in cancer prevention and treatment, people have proposed different mechanisms to explain its anti-cancer effects. More and more data show that vitamin D can regulate the entire process of tumorigenesis, including tumorigenesis, metastasis, and the interaction between cells and the microenvironment. These mechanisms include cell proliferation, differentiation, apoptosis, autophagy, and epithelium- Regulation of interactions between mesenchymal transition, angiogenesis, anti-oxidation, inflammation, and immune system microenvironment. Since the initiation and development stages of tumors are very important in the growth of tumors, this article focuses on the various roles of vitamin D in the initiation of tumors.
Initial Stage: Anti-inflammatory, Anti-oxidant and DNA Damage Repair
The occurrence of tumor is the irreversible gene mutation of normal cells, which in turn induces the transformation of other normal tissues. Studies have shown that vitamin D plays a key anti-tumor effect through anti-inflammatory, anti-oxidant and DNA damage repair during tumor initiation.
Anti-inflammatory Effect
Chronic inflammation can destroy and remodel tissues through reactive oxygen species and cytokines secreted from the site of inflammation. The current accepted view is that chronic inflammation is one of the main causes of tumors. Vitamin D can exert anti-inflammatory effects through at least 4 mechanisms. First, vitamin D inhibits the expression of cyclooxygenase-2 (COX-2) and prostaglandin (PG) receptors in breast cancer cells to achieve the purpose of inhibiting inflammation. Similarly, in prostate cancer cells, vitamin D can also reduce the expression levels of COX-2, PG receptor EP2 and FP. Second, vitamin D can inhibit the pro-inflammatory signaling pathway mediated by p38 mitogen-activated protein kinase (p38 MAPK). In normal prostate epithelial cells and prostate cancer cells, calcitriol inhibits the phosphorylation and activation of p38MAPK by inducing the expression of MAPK phosphatase-5 (MKP-5), thereby inhibiting the production of interleukin-6 (IL-6) and other inflammatory cytokines. Third, vitamin D can also inhibit the NF-κB signaling pathway. In immune cells such as macrophages, B cells and T cells, vitamin D can affect the inflammatory environment of tumor cells by inhibiting the NF-κB signaling pathway. Fourth, vitamin D can regulate the interaction between immune cells and cancer cells and inhibit the production of pro-inflammatory cytokines. Co-culture experiments on peripheral blood mononuclear cells (PBMCs) and colon cancer cells found that vitamin D treatment can significantly reduce the production of pro-inflammatory cytokines, such as tumor necrosis factor (TNF), IL-6 and interleukin-10 (IL- 10), which shows that vitamin D plays an anti-inflammatory effect in the microenvironment of tumor cells.
Antioxidant Defense and DNA Damage Repair
Reactive oxygen species play a key role in many aspects of tumorigenesis through mechanisms such as promoting DNA mutations, inducing cell proliferation, and initiating pro-inflammatory responses. Therefore, maintaining the normal operation of the antioxidant defense system should be a key step to prevent tumor development. Vitamin D not only plays a key role in the body’s anti-inflammatory process, but is also one of the key controllers of oxidative stress and mitochondrial respiratory function. The lack of vitamin D can damage mitochondrial function and aggravate oxidative stress. When the body’s vitamin D is sufficient, many intracellular activities related to oxidative stress are inhibited. However, when the serum 25-OH-D concentration is below a certain value, oxidative stress cannot be inhibited, and the oxidative damage of cells will be aggravated at this time. At the same time, the level of nuclear factor E2-related factor 2 (Nrf2) regulated by vitamin D is negatively correlated with the accumulation of mitochondrial reactive oxygen species. Nrf2 plays a key role in protecting cells from oxidative stress. The results of randomized controlled clinical trials from the population showed that daily supplementation of vitamin D in the human body can reduce DNA oxidative damage, which indicates that vitamin D can protect human DNA from damage caused by oxidative stress. Vitamin D plays a protective role in DNA damage induced by reactive oxygen species, which can be attributed to its participation in the expression of multiple enzymes in this process. Calcitriol can induce the expression of superoxide dismutase 1 (SOD1) and superoxide dismutase 2 (SOD2) in prostate epithelial cells and androgen-sensitive prostate cancer cells (LNCaP), respectively. In addition, calcitriol can induce the expression of thioredoxin reductase 1 (TXNRD1), and TXNRD1 can reduce the antioxidant function of thioredoxin in prostate cancer and breast cancer cells. There are also experiments showing that calcitriol can induce the expression of glucose-6-phosphate dehydrogenase (G6PD), and the reduced coenzyme Ⅱ (NADPH) produced by G6PD can promote glutathione (GSH) in patients with prostate cancer and ovarian cancer. In addition to preventing DNA damage by enhancing the body’s antioxidant capacity, vitamin D can also directly regulate the process of DNA damage repair. Studies have shown that vitamin D increases the expression of genes involved in DNA damage repair, such as p53 in breast cancer cells, proliferating cell nuclear antigen (PCNA), and breast cancer 1 gene (BRCA1). Vitamin D can also inhibit the degradation of p53 binding protein 1 (53BP1) in breast cancer cells. Therefore, vitamin D can prevent gene mutations in the initial stage of tumors through anti-inflammatory, antioxidant, and DNA damage repair functions.
