Triglycerides (TGs) provide the major storage form of fatty acids (FAs), which in turn serve as reservoirs of metabolic fuel ormem-brane building blocks. During times of excess energy availability, more TG is synthesized, and in mammals this TG is predomi-nantly packaged in cellular lipid droplets (LDs), the organelle for neutral lipid storage. TG storage in mammals occurs primarily in adipocytes in white adipose tissue (WAT). However, when adipose depots of TG are over-whelmed, for instance during obesity, other cells will synthesize and accumulate TG in LDs. This is often accompanied by tissue dysfunction due to cellular lipotoxicity, which can lead to lipotoxicity-related diseases, such as type 2 diabetes, non-alcoholic fatty liver disease, and metabolic cardiomyopathy.
For decades, scientists have wondered why some triglycerides, after being broken down into fatty acids, wind up back in the cells in the form of triglycerides — a process known as “re-esterification.” A new study from Harvard T.H. Chan School of Public Health and Howard Hughes Medical Institute sheds light on how a key fat-producing enzyme helps protect cells from a toxic form of fat. The study has been published online August 1, 2017 in Cell Metabolism.
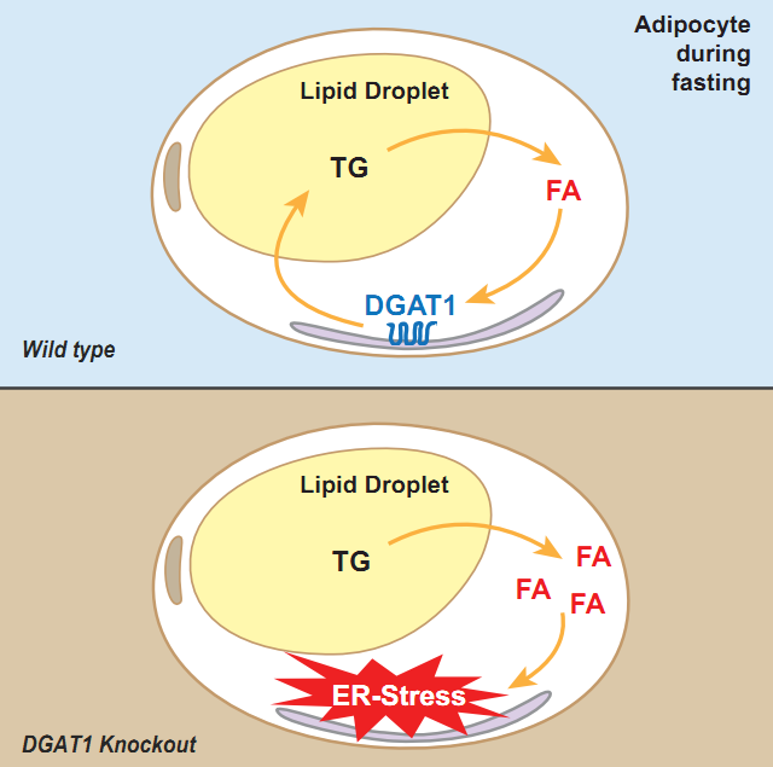
By examining cell processes both in mice and in human cells, the researchers found out why: Re-esterification helps protect a key cell organelle called the endoplasmic reticulum (ER). The ER helps make cellular products such as proteins and lipids and it can be damaged by fatty acids–but not by triglycerides. The researchers also found that an enzyme called DGAT1 (diacylglycerol acyltransferase), an ER-localized DGAT enzyme is crucial to the re-esterification process, fatty acid re-esterification during adipocyte lipolysis is mediated by DGAT1. Surprisingly, this re-esterification cycle does not preserve TG mass but instead functions to protect the ER from lipotoxic stress and related consequences, such as adipose tissue inflammation.
Diacylglycerol O-acyltransferase 1 is an enzyme that in humans is encoded by the DGAT1 gene. This gene encodes an multipass transmembrane protein that functions as a key metabolic enzyme. The encoded protein catalyzes the conversion of diacylglycerol and fatty acyl CoA to triacylglycerol. This enzyme can also transfer acyl CoA to retinol. Activity of this protein may be associated with obesity and other metabolic diseases.
This finding reveals an important role for DGAT activity and TG synthesis generally in averting ER stress and lipotoxicity, with specifically DGAT1 performing this function during stimulated lipolysis in adipocytes. It contributes to a fuller understanding of the fundamental biology that underlies common metabolic diseases related to obesity, such as type 2 diabetes, fatty liver disease, and heart failure, and could lead to new insights on how to better treat such diseases.