Cytomegalovirus (CMV), also known as human cytomegalovirus (HCMV), is a ubiquitous pathogen that infects a large proportion of the global population. In most healthy individuals, infection remains silent and clinically insignificant. However, in specific populations—including newborns, pregnant women, and immunocompromised patients—CMV can lead to severe complications, long-term sequelae, and even life-threatening disease. Understanding how this virus operates at the molecular level, along with how it is accurately detected in clinical settings, is essential for improving outcomes and guiding medical decisions.
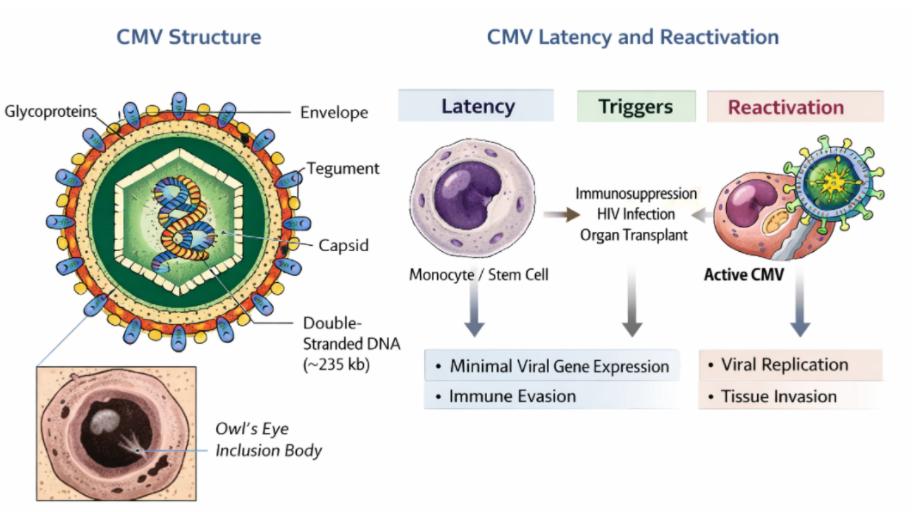
Figure 1. Structural organization and lifecycle dynamics of human cytomegalovirus (CMV).
Understanding CMV as a Persistent Herpesvirus
CMV belongs to the Herpesviridae family, a group of viruses known for their ability to establish lifelong infections after initial exposure. Structurally, CMV contains a large double-stranded DNA genome of approximately 235 kilobases, encoding more than 200 genes. This extensive genetic capacity enables the virus to finely regulate host interactions and adapt to different cellular environments. One of the most recognizable features of CMV infection is the formation of enlarged host cells with prominent intranuclear inclusions, commonly described as “owl’s eye” appearances under microscopy, which reflect active viral replication within the nucleus.
Molecular Basis: Latency and Reactivation
A defining characteristic of CMV is its ability to transition between latent and active states. After primary infection, the virus does not get completely eliminated. Instead, it establishes latency within specific cell types, particularly monocytes, macrophages, and hematopoietic progenitor cells in the bone marrow. During latency, viral gene expression is highly restricted, allowing the virus to remain largely invisible to the host immune system.
Reactivation occurs when immune surveillance is compromised. Conditions such as HIV infection, organ transplantation, immunosuppressive therapy, or advanced age can disrupt immune control, allowing the virus to resume replication. Once reactivated, CMV can spread systemically and invade multiple organs, leading to clinical manifestations such as pneumonia, gastrointestinal disease, or central nervous system involvement. This balance between latency and reactivation is central to CMV persistence and explains why the virus poses a greater threat in vulnerable populations.
Immune Evasion and Host Interaction
CMV has evolved a range of sophisticated strategies to evade immune detection and establish long-term persistence. One of its primary mechanisms involves interference with antigen presentation pathways. By disrupting the expression and transport of major histocompatibility complex (MHC) class I molecules, CMV prevents infected cells from being recognized and destroyed by cytotoxic T lymphocytes.
In addition, CMV targets innate immune sensing systems, particularly the cGAS-STING pathway, which is normally responsible for detecting foreign DNA within the cell. By suppressing this pathway, the virus reduces the host’s ability to initiate antiviral responses. CMV also modulates the host inflammatory environment by influencing cytokines such as interleukin-1 and tumor necrosis factor-alpha, as well as altering chemokine signaling and adhesion molecules. These changes not only facilitate viral persistence but can also contribute to immune dysregulation and tissue damage during active infection.
Diagnostic Workflow: The Importance of Timing and Sample Selection
Accurate diagnosis of CMV infection relies heavily on selecting the appropriate sample type and collecting it within the correct time frame. This is particularly critical in cases of congenital CMV infection, where distinguishing between infection acquired before birth and after delivery has significant clinical implications. The first 21 days of life represent the optimal diagnostic window. Beyond this period, it becomes difficult to determine whether the infection is congenital or postnatal.
Among available sample types, urine is considered the most reliable due to its high viral load, offering excellent sensitivity for detection. Saliva is also widely used because of its convenience and non-invasive collection, although confirmatory testing is often recommended to rule out contamination. Dried blood spots provide an additional option, especially in retrospective cases where early samples are unavailable, although their sensitivity is comparatively lower.
Laboratory Methods: From Classical to Molecular Diagnostics
The evolution of CMV diagnostics has significantly improved detection accuracy and clinical decision-making. Polymerase chain reaction (PCR) has become the cornerstone of modern CMV testing. By directly detecting viral DNA, PCR offers both high sensitivity and specificity, along with the ability to quantify viral load. This quantitative aspect is particularly valuable in monitoring disease progression and treatment response in immunocompromised patients.
Serological testing, which measures CMV-specific antibodies such as IgM and IgG, plays a more limited role. While IgM may suggest recent infection, it can persist for extended periods and sometimes yield false-positive results. IgG, on the other hand, indicates prior exposure but does not provide information about the timing or activity of infection. As a result, serology is generally used as a complementary tool rather than a primary diagnostic method.
Historically, antigen detection methods such as the pp65 antigenemia assay were used to identify CMV proteins in circulating leukocytes. However, these approaches have largely been replaced by PCR due to their lower sensitivity and greater technical complexity.
Clinical Interpretation and Disease Stratification
Interpreting CMV test results requires careful consideration of both laboratory findings and clinical presentation. In congenital CMV infection, infants may be asymptomatic at birth yet still face a measurable risk of developing long-term complications, particularly sensorineural hearing loss. Symptomatic cases, by contrast, often present with more obvious signs such as jaundice, hepatosplenomegaly, or neurological abnormalities, reflecting more extensive viral involvement.
In immunocompromised individuals, the presence of a high viral load combined with compatible clinical symptoms is indicative of invasive CMV disease. This can manifest in various forms, including pneumonia, encephalitis, and retinitis, depending on the organs affected. Early recognition and monitoring are essential to prevent disease progression and guide antiviral therapy.
Post-Diagnostic Evaluation and Clinical Management
Once CMV infection is confirmed, further evaluation is necessary to assess the extent of disease and determine appropriate management strategies. Hearing assessment is particularly important in newborns, as CMV is a leading cause of non-genetic congenital hearing loss. Neuroimaging, especially magnetic resonance imaging, provides valuable insights into potential central nervous system involvement and is more sensitive than ultrasound for detecting structural abnormalities.
Ophthalmologic examination is also recommended to identify retinal involvement, which may lead to visual impairment if left untreated. In addition, routine laboratory tests such as complete blood counts can reveal hematological abnormalities that may influence clinical decisions.
Conclusion
CMV represents a highly adaptable virus that has evolved to coexist with its host through mechanisms of latency and immune evasion. While it remains largely benign in healthy individuals, its impact on vulnerable populations underscores the importance of early detection and accurate diagnosis. Advances in molecular diagnostics, particularly PCR-based methods, have transformed the clinical approach to CMV, enabling timely identification and improved patient management. A comprehensive understanding of both the molecular basis of infection and the diagnostic workflow is essential for clinicians, researchers, and healthcare professionals working to mitigate the burden of this widespread virus.
References
1. Nguyen AD, Shorman M. Cytomegalovirus Infections. 2025 Dec 13. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. 2026, PMID: 29083720.
2. Kampouri E, et al.; Cytomegalovirus (CMV) Reactivation and CMV-Specific Cell-Mediated Immunity After Chimeric Antigen Receptor T-Cell Therapy. Clin Infect Dis. 2024, 78(4):1022-1032.
Related Products
| Pathogen | Cat. No. | Product Name | Isotype | Application | |
| CMV | DMAB-CBXY25310 | Human Anti-CMV gB Mab, clone 22G | IgG | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25311 | Human Anti-CMV gB Mab, clone 22M | IgM | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25312 | Human Anti-CMV gH/gL Mab, clone 23G | IgG | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25313 | Human Anti-CMV gH/gL Mab, clone 23M | IgM | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25316 | Human Anti-CMV Mab, clone 70C15G | IgG | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25317 | Human Anti-CMV Mab, clone 70C15M | IgM | ELISA, Control | Inquiry |
| CMV | DMAB-CHD25046 | Human Anti-CMV pp150 Mab, clone 2B21 | IgG1 | ELISA, Control | Inquiry |
| CMV | DMAB-CHD25047 | Human Anti-CMV pp150 Mab, clone 3B33 | IgM | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25314 | Human Anti-CMV pp65 Mab, clone 67R23G | IgG | ELISA, Control | Inquiry |
| CMV | DMAB-CBXY25315 | Human Anti-CMV pp65 Mab, clone 67R23M | IgM | ELISA, Control | Inquiry |
