Immune cell infiltration of solid tumors is one of the hallmarks of cancer and plays a vital role in disease progression. Through the recognition of the important role of immune cells in cancer, immunotherapy strategies have been introduced. Cells of the myeloid lineage are the main cell source for tumor immune cell infiltration; however, current immunotherapy only involves the adaptive immune system. The tumor microenvironment may reprogram immune cells that infiltrate the tumor, thereby changing their properties to promote tumor growth. What’s more noteworthy is that cancer can cause changes in bone marrow production, which in turn promotes the increase in the production of myeloid cells including monocytes, macrophages and neutrophils. These cells then accumulate on the tumor, and Usually move towards promoting a specific phenotype of the tumor. However, the process of tumor-infiltrating myeloid cells is complex and dynamic, and certain tumor types and innate immune cells may also be transformed to continue to exert anti-tumor activity. Therefore, immunotherapy aimed at reversing the pre-tumor phenotype in innate immune cells or promoting its anti-tumor phenotype represents an attractive anti-tumor method that can work synergistically with current immunotherapies for adaptive immunity.
Existing clinical data indicate that fungal-derived polysaccharide β-glucan or Bacillus Calmette-Guerin-Bacillus vaccine (BCG) can promote the continuous enhanced response of bone marrow cells to secondary infection or inflammation. The process mediated by transcription, epigenetic and metabolic reprogramming is called “trained innate immunity” or “innate immune memory”. Recently, scientists have discovered that by regulating the progenitor cells of myeloid cells in the bone marrow (BM), they have discovered that the trained immune system can exhibit long-term anti-cancer effects. In addition, agonist agents that help train the immune system exert anti-tumor activity. For example, BCG can be used to treat bladder cancer, and β-glucan is related to the efficacy of tumor immunotherapy. However, its specific role in tumors is still not clear. It is not well understood whether the potential tumor-modulating effects of agents such as β-glucan involve the induction of innate immune memory. In order to clarify the mechanism, scientists have recently explored the induction experiment by pretreatment of mice with β-glucan.
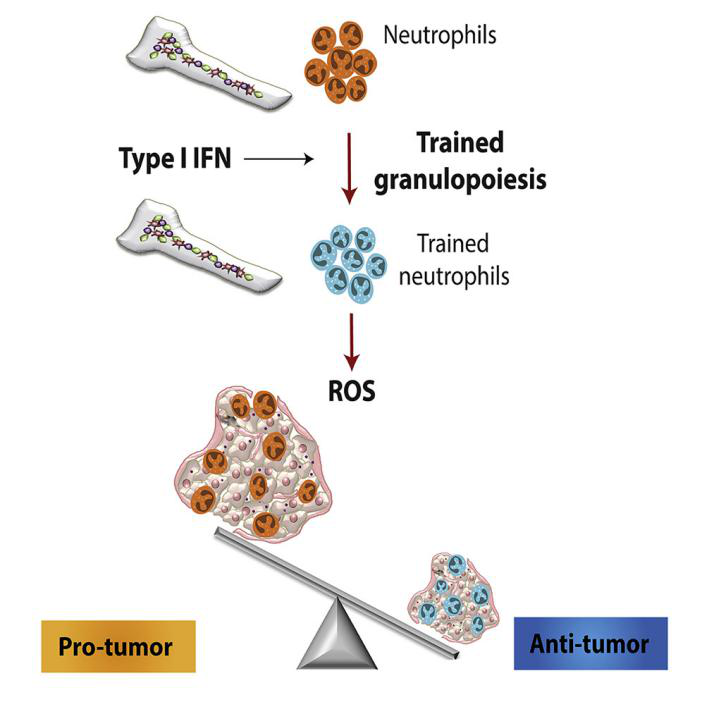
Studies have shown that neutrophils infiltrating solid tumors (called tumor-associated neutrophils (TANs)) can exhibit phenotypes with cytotoxic and anti-tumor properties (widely referred to as TAN1) or with Phenotype related to tumor progression (TAN2). Type I interferon (IFN) promotes TAN1, while transforming growth factor-β (TGF-β) is related to the differentiation of TAN2. In the context of anti-tumor immunity, neutrophils and granulocyte progenitor cells are the main cellular effectors of training immunity induced by β-glucan. Further studies have found that the anti-tumor effect of training immunity induced by β-glucan is related to the transcriptome and epigenetic rearrangement of granulocyte production, as well as neutrophil reprogramming and anti-tumor phenotype. This process is related to granulocyte production mediated by type I IFN signaling. Studies have shown that type I interferon plays a central role in granulocyte production and promotes the anti-tumor phenotype of neutrophils. Mice lacking IFN-β showed hindered bone marrow hematopoietic progenitor cell maturation, reduced blood neutrophil counts, and more aggressive tumor growth. After immune training, neutrophils can achieve subsequent anti-tumor properties through the generated ROS. The results showed that the adoptive transfer of neutrophils from β-glucan-trained mice to naive receptors inhibited the tumor growth of the latter in a ROS-dependent manner. Therefore, the anti-tumor effect of the trained granulocytes induced by β-glucan can be transmitted to unimmunized mice through bone marrow transplantation.
