The Hidden Risk Behind Biological Drug Manufacturing
In te world of biopharmaceuticals, impurity testing is rarely just a technical task—it sits at the intersection of patient safety, regulatory compliance, andproductintegrity. Among the many impurities that must be controlled, antibiotic residues remain particularly important. These compounds are widely used during cell culture, viral amplification, and upstream processing, yet even trace levels can persist into the final product if not carefully monitored.
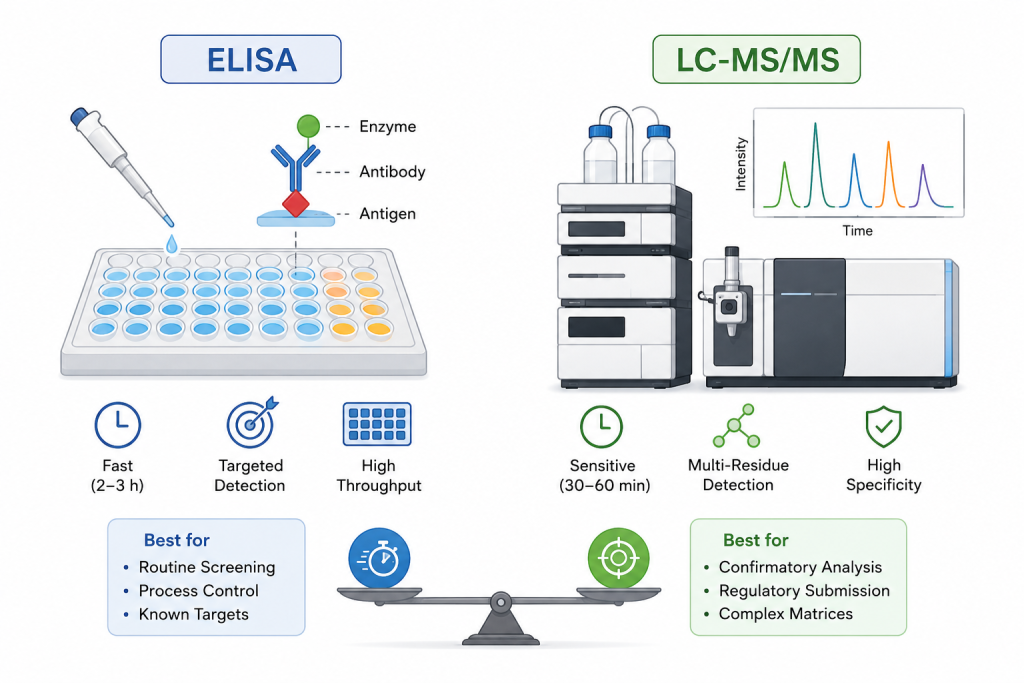
From vaccines to monoclonal antibodies and gene therapies, manufacturers face the same question: how do you detect antibiotic residues with enough sensitivity, speed, and reliability to satisfy both scientific and regulatory expectations? In practice, this decision often comes down to choosing between two established analytical approaches—Enzyme-Linked Immunosorbent Assay and Liquid Chromatography–Tandem Mass Spectrometry—each with its own philosophy, strengths, and trade-offs.
Where Do Antibiotic Residues Come From?
Antibiotic residues enter biological products through necessary but tightly controlled steps. For example, aminoglycosides like neomycin or gentamicin are frequently used in vaccine production to prevent microbial contamination during cell culture. Similarly, penicillin or ampicillin may be introduced in recombinant protein or monoclonal antibody workflows.
While these agents serve a critical role during manufacturing, their unintended persistence can have serious consequences. Even low-level residues may trigger allergic reactions, contribute to antimicrobial resistance, or interfere with the biological activity of the drug. This is why regulatory authorities, including the International Council for Harmonisation, impose strict limits and require validated detection methods capable of operating at very low concentration thresholds.
ELISA: Speed and Simplicity for Routine Screening
Enzyme-Linked Immunosorbent Assay has become a cornerstone of routine impurity testing, particularly in high-throughput environments. Its popularity stems from a straightforward principle: antigen–antibody specificity. This allows laboratories to rapidly screen for known antibiotic residues with relatively simple workflows.
In practice, ELISA excels when time and scalability are critical. A single analyst can process dozens or even hundreds of samples in parallel using microplate formats, often obtaining results within a few hours. This makes it especially valuable for in-process monitoring, where rapid feedback helps maintain control over manufacturing conditions.
However, ELISA’s reliance on antibodies also introduces limitations. The quality and specificity of the antibody determine the assay’s accuracy, and structurally similar compounds can sometimes cause cross-reactivity. Additionally, ELISA is inherently targeted—each assay is designed for a specific analyte. When multiple or unknown residues are involved, its flexibility becomes limited.
LC-MS/MS: Precision and Depth for Confirmatory Analysis
By contrast, Liquid Chromatography–Tandem Mass Spectrometry offers a fundamentally different analytical approach. Instead of relying on biological recognition, it identifies compounds based on their molecular structure and mass-to-charge ratio. This enables exceptionally high sensitivity and specificity, often reaching detection limits in the ng/mL range.
In complex biological matrices, such as monoclonal antibody formulations or gene therapy products, this capability becomes indispensable. LC-MS/MS can separate and quantify multiple antibiotic residues within a single run, reducing ambiguity and providing highly reliable data for regulatory submissions.
That said, this level of performance comes with increased complexity. The instrumentation is costly, sample preparation can be labor-intensive, and method development requires technical expertise. For routine screening of large sample volumes, these factors may reduce operational efficiency.
Why Many Labs Use Both Instead of Choosing One
Given their complementary strengths, many laboratories no longer view ELISA and LC-MS/MS as competing options. Instead, they are integrated into a tiered analytical strategy.
ELISA is typically used as a first-line screening tool, enabling rapid and cost-effective monitoring during production. Samples that require further verification are then analyzed using LC-MS/MS, which provides the accuracy and sensitivity needed for confirmatory testing and final product release.
This combined approach not only improves efficiency but also aligns with regulatory expectations for orthogonal methods. By using two fundamentally different techniques, manufacturers can strengthen data reliability and reduce the risk of false results.
Matching the Method to the Product
The choice of analytical method often depends on the type of biological product and its complexity. Vaccines, for instance, frequently rely on ELISA for routine monitoring of residues like neomycin, where high throughput is essential. In contrast, monoclonal antibodies and gene therapy products, which involve more complex matrices and stricter regulatory scrutiny, are more likely to require LC-MS/MS for accurate quantification.
In early-stage research and development, LC-MS/MS is often preferred because of its ability to screen multiple residues simultaneously. During manufacturing, ELISA provides a practical solution for ongoing process control. At the final release stage, LC-MS/MS ensures that products meet stringent regulatory standards.
The Critical Role of Sample Preparation
Regardless of the analytical platform, sample preparation remains a decisive factor in data quality. Techniques such as ultrafiltration, solid-phase extraction, and immunoaffinity purification are commonly used to remove interfering substances and concentrate target analytes.
Poor sample preparation can compromise both ELISA and LC-MS/MS results—leading to false positives in immunoassays or signal suppression in mass spectrometry. As a result, robust and validated preparation protocols are essential for reliable impurity testing.
Looking Ahead: Smarter and Faster Detection Technologies
The field of impurity testing is evolving rapidly. Advances in automation are making ELISA even more efficient, while innovations in mass spectrometry continue to push the boundaries of sensitivity and speed. At the same time, emerging technologies such as biosensors, microfluidic systems, and AI-assisted data analysis are opening new possibilities for real-time and high-throughput detection.
These developments suggest a future where impurity testing becomes more integrated into the manufacturing process itself, enabling earlier intervention and better control over product quality.
Conclusion: It’s About Strategy, Not Just Technology
Choosing between ELISA and LC-MS/MS is not about identifying a universally superior method. Instead, it is about selecting the right tool for the right context. ELISA offers speed, simplicity, and scalability, making it ideal for routine monitoring. LC-MS/MS provides unmatched sensitivity and analytical depth, ensuring confidence in complex and high-stakes analyses.
For most organizations, the optimal solution lies in combining both techniques into a cohesive strategy. By doing so, they can meet regulatory expectations, improve operational efficiency, and ultimately ensure that biological drugs are as safe and effective as possible before reaching patients.
References
1.Pilely K, et al.; Monitoring process-related impurities in biologics-host cell protein analysis. Anal Bioanal Chem. 2022, 414(2):747-758.
Related Products
| Cat. No. | Product Name | Sample | |
| DEIA040 | Ampicillin | Vaccine | Inquiry |
| DEIABL-QB25 | Benzyl penicillin | Biological samples | Inquiry |
| DEIA6881 | Chloramphenicol | Biological samples, fish, shrimp, eggs, honey, meat (beef, chicken and pork), milk, milk powder, condensed milk and serum | Inquiry |
| DEIA6881V2 | Chloramphenicol | Vaccine | Inquiry |
| DEIA042 | Colistin | Beef, pork, chicken | Inquiry |
| DEIABL-QB20 | Erythromycin | Tissue(muscle), cell supernatant, vaccine, water, honey, milk | Inquiry |
| DEIA047 | Gentamicin | Vaccine, cell culture supernatant | Inquiry |
| DEIA-WZ6884 | High Sensitivity Gentamicin | Cell lysate, urine, serum, recombinant protein, fermentation liquid | Inquiry |
| DEIA048 | Kanamycin | Biological samples | Inquiry |
| DEIA-WZ048V | High Sensitivity Kanamycin | Biological samples | Inquiry |
| DEIA043 | Neomycin | Cell culture supernatant, vaccine, milk | Inquiry |
| DEIA-XYZ35 | Oxytetracycline | Biological samples, honey, butter, fermented milk, feed, egg, cheese, milk, meat, meat products (sausage), fish and shrimp | Inquiry |
| DEIA020 | Streptomycin | Vaccine, cell culture supernatant | Inquiry |
| DEIA-WZ020 | High Sensitivity Streptomycin | Serum, urine, recombinant protein | Inquiry |
| DEIANJ11 | Human Vancomycin | Human serum, plasma | Inquiry |
