As a specific marker of synaptic terminals, synaptic proteins, when combined with receptors, exert biological functions and can be applied to the study of nervous system development, disease and plasticity. At present, many studies have found changes in synaptic protein expression in neurological diseases such as cerebral ischemia and hypoxia, Alzheimer's disease, prion disease and epilepsy. In-depth study of changes in synaptic proteins in the nervous system can provide a good means for studying its pathogenesis, pathological and physiological changes, providing new ideas for the prevention and treatment of certain neurological diseases, as well as providing a reliable basis for treatment method.
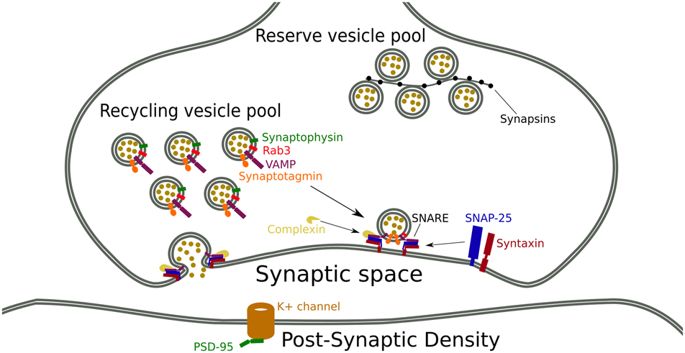
Synaptic proteins have two main functions: regulation of neurotransmitter release and participation in the early development of neurons. In regulating the release of neurotransmitters, synaptic proteins can maintain the stability of the synaptic vesicle reservoir. Presynaptic vesicles are distributed in two pools, one containing a small number of vesicles, anchored in the active zone, ready to release neurotransmitters, called the release pool; the other pool contains most of the vesicles, away from the active zone, called the storage pool. The reservoir has enough vesicles to ensure that the synapse responds to high levels of neural activity. At rest, synaptic proteins reversibly link vesicles to vesicles and attach vesicles to the cytoskeletal network formed by troponin, thereby leaving vesicles away from the plasma membrane; neurotransmitters are released thereafter; synaptic proteins can cause the renewed vesicles to return to the reservoir, thereby maintaining the dynamic stability of the vesicle reservoir. In synaptic protein I and II knockout mice, a selective decrease in the number of bubbles in the synaptic reservoir were observed, suggesting that synaptic proteins may play an important role in maintaining the stability of the vesicle reservoir. Regulating the mobility of synaptic vesicles: According to the vesicle activity hypothesis, when the concentration of Ca2+ in the cytoplasm near the active zone is increased, CaMKII is activated, which phosphorylates synapsin-I, which makes it the same as troponin. The ability of the protein to bind is weakened, increasing the mobility of the vesicles. As the research progresses, the hypothesis is constantly being supplemented. Chi et al. provide direct physiological evidence for synaptic protein regulation of vesicle activity. At the end of the axon of living hippocampal neurons, synaptic protein-I labeled with green fluorescent protein can be observed when action potentials are generated. Synaptic vesicles separate and diffuse toward the axons, and re-aggregate to the synapses when synaptic activity ceases. At the same time, by blocking the phosphorylation of various sites, it was found that the diffusion rate of synaptic proteins is controlled by phosphorylation. Phosphorylation can be caused by a variety of PKs acting on specific phosphorylation sites of synaptic proteins, while phosphorylation at different sites produces different effects. Taking synapsin-I as an example, locus 1 is the site of action of PKA, and sites 2 and 3 are sites of action of CaMK-II, and phosphorylation of these three sites causes synapsin-I and vesicles decrease the affinity and block the interaction between synapsin-I and troponin. Phosphorylation sites 4, 5, and 6 are the sites of action of MAPK, and phosphorylation inhibits the interaction between synapsin-I and troponin but has no effect on its affinity with vesicles. Regulation of kinetics of neurotransmitter release: Recent studies have found that synaptic proteins regulate the release of independent vesicles from the release cell. Hilfiker et al. injected the polypeptide fragment of the E region by the giant presynaptic squid. It was observed that the number of vesicles in the release cell did not change, but the release process of the neurotransmitter was inhibited and slowed down, suggesting that the synaptic protein may anchor on the vesicle to regulate the release of neurotransmitters. Hilfiker also supported the above speculation by the delay in the occurrence and rise of excitatory post-synaptic currents in squid. Several evidences have shown that the slowing down of the kinetics of this neurotransmitter release is neither a result of a decrease in transmitter release nor an overall decrease in synaptic vesicle pools. Weber et al. suggest that the kinetic mechanism of synaptic protein regulation of neurotransmitter release may be: E region may play a regulatory role in the correct location of vesicles and the binding of synaptic proteins to vesicles, and it may be the necessary factors to ensure the correct positioning and fusion of the vesicles. Regulating synaptic plasticity: In recent years, the relationship between synaptic proteins and synaptic plasticity has received attention because of its potential to be associated with complex learning and memory functions and neurological rehabilitation of the nervous system. Humeau et al. injected a specific anti-synaptic antibody into a mollusk to neutralize endogenous synaptic proteins and found that the synaptic tonic post-enhancement (PTP) disappeared and was replaced by tonic (PTD), while synaptic failure speeds up and increases. This suggests that synaptic proteins may be involved in the regulation of short-term plasticity in synapses. Sato et al. used quantitative immunohistochemical analysis to find synaptic-I immunopositive substances in the dentate gyrus of rats and the transparent layer of the hippocampal CA3 region within 8h after stimulation to produce long-term potentiation (LTP). Participation in the early development of central neurons: regulation of axon and dendritic elongation studies suggests that synaptic proteins have been expressed during the early developmental stages of neurons and synapse formation, which is mainly involved in elongation. The cell region, in which synaptic protein-III is most expressed, is concentrated in the growth cone. Leypoldt et al used retinoic acid to promote differentiation of human NTERA-2clD1 cells and found that the transcriptional levels of three members of synaptic proteins were significantly up-regulated, with synaptic protein-I increased by 2.1 times, synaptic protein-II increased by 2.6 times, while the synaptic protein-III increased by 5. 5 times. The absolute advantage of synapsin-III suggests an irreplaceable role in early presynaptic development. The process of elongation of axons and dendrites is divided into four stages: the neurons are partially or surrounded by lamellae; lobes form the same short protrusions; one of the small protrusions begins to elongate rapidly and axons are formed; the remaining small protuberances also elongate and differentiate into dendrites, and inhibition of expression of different members of synaptic proteins can lead to changes in different stages. Previous studies have found that hippocampal neurons obtained from synapsin-I knockout mice can differentiate between axons and several small dendrites, but the length of the axis and dendrites is significantly shorter than normal. Recent studies have found that inhibition of synapsin-II expression results in 20% of neurons failing to grow protuberances, 80% of malformed protuberances, and these malformed protuberances are inseparable during the first 48 h of culture. Hippocampal neurons lacking synaptic protein-III can grow normal processes but fail to elongate and differentiate into axons and dendrites. Participation in the formation and maintenance of synapses: Many studies have shown that synaptic proteins are involved in the process of synapse formation. The synaptic terminals were observed to be smaller in synapsin-I knockout mice, and the number of synaptic vesicles was also significantly reduced. Ferreira et al. extracted hippocampal neurons from mice with synaptic protein mutations and found that although hippocampal neurons can form synaptic connections, the synaptic connections formed by normal controls are significantly reduced. The occurrence of the touch was delayed by nearly 1 week. Synaptic proteins are involved in the formation of synapses mainly through two aspects: On the one hand, the distribution of synaptic vesicles can be adjusted. On the other hand, synaptic proteins can also be involved in the regulation of other synapse-associated proteins. Inhibition of expression of synapsin-I or II can result in a decrease in the expression of multiple presynaptic proteins. It is worth noting that synaptic protein-III is not involved in the formation of synaptic connections, and inhibition of synapsin-III expression after axonal elongation does not affect the process of synapse formation. In addition to participating in the formation of synapses, synaptic proteins are also involved in the maintenance of synapses. Using antisense nucleotide technology to inhibit the expression of synapsin-II, Ferreira et al. found that inhibition of synaptic protein-II expression in hippocampal neurons before synaptic formation leads to inhibition of synaptic development. Restraining the expression of synaptic connections leads to the loss of most synapses by neurons.
There are five synaptic proteins that have been discovered, synapsin-Ia, synapsin-Ib, synapsin-IIa, synapsin-IIb, and synapsin-IIIa. The touch protein is distributed in dots at all nerve terminals. In the central nervous system, synaptic proteins are mainly distributed in the brain, spinal cord, and retina, and no staining domain of white matter. In the peripheral nervous system, mainly in the nerve and muscle junctions and in the autonomic ganglia, each synaptic protein is composed of several different domains. A total of 10 different protein domains (A to J regions) have been identified, of which regions A, B, and C are domains of each synaptic protein, and regions A and C are highly conserved domains. The E region is a domain in which the subtype synaptic proteins share different domains and their functions are different. Since different synaptic proteins contain both the same domain and different domains, different synaptic proteins have the same function and different functions. The A region is located at the N-terminus of the synaptic protein and contains the only conserved phosphorylation site, protein kinase A (PKA) and the phosphorylation site of Ca2+/CaM-dependent protein kinase I (CaMKI). In the non-phosphorylated state, synaptic proteins may bind to synaptic vesicles in the form of dimers via phospholipid binding activity associated with region A. Phosphorylation of region A inhibits the binding of synaptic proteins to the vesicle phospholipid membrane, resulting in the release of synaptic proteins from synaptic vesicles. Area C is the largest and conserved domains at the heart of synaptic proteins. Area E is a consensus domain of synaptic protein isoforms, whose main function is to regulate the stability and kinetics of release of neurotransmitters in the synaptic vesicle reservoir. Protein kinase (MAPK) phosphorylation acts primarily to connect adjacent structures and participates in the regulation of the release of neurotransmitters. The function of the synaptic protein is dependent on the binding of synaptic proteins and synaptic receptors. More studies are now about synaptic protein blockers, which can block some of the functions of synaptic proteins. The specific mechanism is still under further research, and there is currently no drug for clinical treatment.
References: