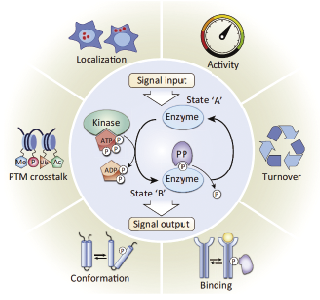
Reversible protein phosphorylation, principally on serine, threonine or tyrosine residues, is one of the most important and well-studied post-translational modifications. Phosphorylation plays critical roles in the regulation of many cellular processes including cell cycle, growth, apoptosis and signal transduction pathways. Phosphorylation is the most common mechanism of regulating protein function and transmitting signals throughout the cell. After the development of Phosphor-Specific Antibody, ELISA has become a powerful method for measuring protein phosphorylation.
Creative diagnostics provides high quality phosphorylation ELISA kits to deliver accurate, sensitive, and fast protein phosphorylation events detection in a broad range of sample types.
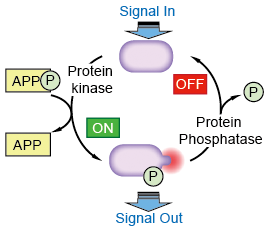 Figure 1. Protein phosphorylation is a
Figure 1. Protein phosphorylation is a
very common means of regulating protein activity.
A method commonly used by eukaryotic cells to regulate protein activity is attaching a phosphate group covalently to one of its amino acid side chains. Because each phosphate group carries two negative charges, the enzyme-catalyzed addition of a phosphate group to a protein can cause a major conformational change by, for example, attracting a cluster of positively charged amino acid side chains. This conformational change can, in turn, affect the binding of ligands elsewhere on the protein surface-thus altering the protein's activity. Removal of the phosphate group by a second enzyme returns the protein to its original conformation and restores its initial activity. This reversible protein phosphorylation controls the activity of many different types of proteins in eukaryotic cells; in fact, this method is used so extensively that more than one-third of the 10,000 or so proteins in a typical mammalian cell appear to be phosphorylated at any one time. The addition and removal of phosphate groups from specific proteins often occurs in response to signals that specify some change in a cell's state. For example, the complicated series of events that takes place as a eukaryotic cell divides is timed in this way. And many of the signals generated by hormones and neurotransmitters are carried from the plasma membrane to the nucleus by a cascade of protein phosphorylation events.
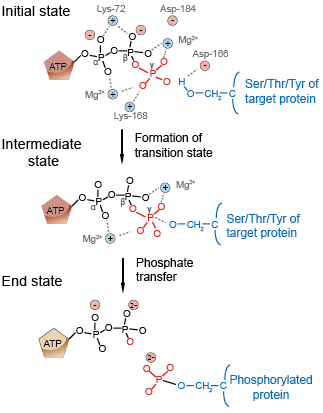 Figure 2. Mechanism of phosphorylation by protein kinase A.
Figure 2. Mechanism of phosphorylation by protein kinase A.
Protein phosphorylation involves the enzyme-catalyzed transfer of the terminal phosphate group of ATP to the hydroxyl group on a serine, threonine, or tyrosine side chain of the protein. This reaction is catalyzed by a protein kinase. The reverse reaction-removal of the phosphate group, or dephosphorylation-is catalyzed by a protein phosphatase.(Figure 1.) Phosphorylation can either stimulate protein activity or inhibit it, depending on the protein involved and the site at which it is being phosphorylated.
To illustrate how the active site binds a specific substrate and then promotes a chemical change in the bound substrate, we examine the action of cyclic AMP-dependent protein kinase, now generally referred to as protein kinase A (PKA). This enzyme and other protein kinases, which add a phosphate group to serine, threonine, or tyrosine residues in proteins, are critical for regulating the activity of many cellular proteins, often in response to external signals. After substrates have bound and the catalytic core of protein kinase A has assumed the closed conformation, the phosphorylation of a serine or threonine residue on the target peptide can take place (Figure 2). As with all chemical reactions, phosphoryl transfer catalyzed by protein kinase A proceeds through a transition state in which the phosphate group to be transferred and the acceptor hydroxyl group are brought into close proximity. Binding and stabilization of the intermediates by protein kinase A reduce the activation energy of the phosphoryl transfer reaction, permitting it to take place at measurable rates under the mild conditions present within cells. Formation of the products induces the enzyme to revert to its open conformational state, allowing ADP and the phosphorylated target peptide to diffuse from the active site.