Notch proteins are a family of type-1 transmembrane proteins that form a core component of the notch signaling pathway, which is highly conserved in metazoans. Notch proteins appear to act as transmembrane receptors for intercellular signals that specify cell fates during animal development. Notch receptors are single-pass, transmembrane proteins conserved from C.elegans to humans. In vertebrates there are four known notch receptors: notch1, notch2, notch3 and notch4. Notch receptors are heterodimeric proteins, consisting of a large, modular N-terminal extracellular portion non-covalently bound to a transmembrane domain, followed by an intracellular, C-terminal portion.
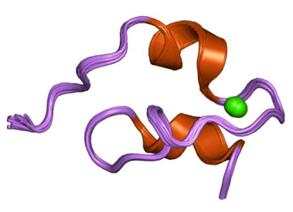 Figure 1. Structure of a Prototype LNR Module from Human Notch1.
Figure 1. Structure of a Prototype LNR Module from Human Notch1.
Table 1. Notch family related products
Notch homolog 1, translocation-associated (Drosophila), also known as NOTCH1, is a human gene encoding a single-pass transmembrane receptor. This gene encodes a member of the Notch family. Notch 1, when activated before birth, induces radial glia differentiation, but postnatally induces the differentiation into astrocytes. One study shows that Notch-1 cascade is activated by Reelin in an unidentified way. Reelin and Notch1 cooperate in the development of the dentate gyrus, according to another.
Neurogenic locus notch homolog protein 2 also known as notch 2 is a protein that in humans is encoded by the NOTCH2 gene. Notch 2 is a member of the notch family. Notch 2 is associated with Alagille syndrome and Hajdu–Cheney syndrome. Mutations within the last coding exon of Notch 2 that remove the PEST domain and escape the nonsense-mediated mRNA decay have been shown to be the main cause of the Hajdu-Cheney syndrome.
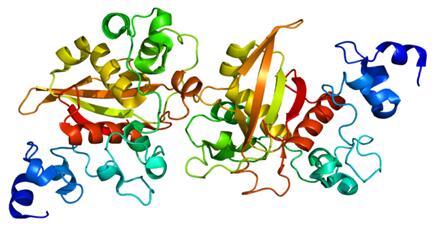 Figure 2. Structure of the Notch2 protein.
Figure 2. Structure of the Notch2 protein.
Neurogenic locus notch homolog protein 3 is a protein that in humans is encoded by the NOTCH3 gene. This gene encodes the third discovered human homologue of the Drosophilia melanogaster type I membrane protein notch. In Drosophilia, notch interaction with its cell-bound ligands (delta, serrate) establishes an intercellular signalling pathway that plays a key role in neural development.
Neurogenic locus notch homolog 4 also known as notch 4 is a protein that in humans is encoded by the NOTCH4 gene located on chromosome 6. Notch 4 protein is a member of the Notch family. An alternative splice variant of the NOTCH4 gene has been described, but its biological significance has not been determined. Mutations in Notch 4 gene may be associated with susceptibility to schizophrenia in a small portion of cases.
Members of this Type 1 transmembrane protein family share structural characteristics including an extracellular domain consisting of multiple epidermal growth factor-like (EGF) repeats, and an intracellular domain consisting of multiple, different domain types. Notch family members play a role in a variety of developmental processes by controlling cell fate decisions. The Notch signaling network is an evolutionarily conserved intercellular signaling pathway that regulates interactions between physically adjacent cells.
In Drosophila, notch interaction with its cell-bound ligands (delta, serrate) establishes an intercellular signaling pathway that plays a key role in development. This protein functions as a receptor for membrane bound ligands, and may play multiple roles during development. A deficiency can be associated with bicuspid aortic valve. Homologues of the notch-ligands have also been identified in humans, but precise interactions between these ligands and the human notch homologues remain to be determined. This protein is cleaved in the trans-Golgi network, and presented on the cell surface as a heterodimer. This protein functions as a receptor for membrane bound ligands, and may play multiple roles during development.
There is evidence that activated Notch 1 and Notch 3 promote the differentiation of progenitor cells into astroglia. Notch 1, then activated before birth, induces radial glia differentiation, but postnatally induces the differentiation into astrocytes. One study shows that Notch 1 cascade is activated by Reelin in an unidentified way. Reelin and Notch 1 cooperate in the development of the dentate gyrus, according to another.
The notch protein family is a critical regulator of differentiation programs. Its ability to affect the cell cycle kinetics and the response to apoptotic signals suggest that notch proteins may be involved in the malignant transformation of some cell systems. In the last decade evidence has accumulated casting light on notch participation in carcinogenesis and human tumors.
Notch activation has been mainly associated to its multiple effects in sustaining oncogenesis, including the control of tumor cell proliferation, migration, cell cycle progression, and inhibition of apoptosis. Indeed, Notch signaling is constitutively activated in several types of cancer cells and it is generally regarded as an antiapoptotic and pro-oncogenic signal. Notch3 overexpression has been reported to be responsible for increased in vitro tumor cell growth in human lung cancer and Notch3 constitutive activation was reported to inhibit terminal differentiation in lungs of transgenic mice. Moreover, increased expression of Notch3 has been observed in spontaneous human pancrea tumors and in T-ALL. In addition to maintaining precursor cells, Notch signaling also participates in the induction of terminal differentiation and growth arrest.
References: