With the demand for vaccine development, Creative Diagnostics have developed two ELISA kits for qualitative detection of SARS-CoV-2 neutralizing antibody in serum and plasma.
Our competitive assay format measures neutralizing antibodies of the SARS-CoV-2 spike RBD and ACE2 interaction in human plasma and serum. For DEIASL055, recombinant human ACE2 receptor protein is immobilized on the plate, followed by the addition of neutralizing antibody or HRP labelled RBD. This interaction is then detected by reading absorbance at 450 nm. The interaction between SARS-CoV-2 spike RBD and ACE2 is disrupted in the presence of neutralizing antibodies in the sample, preventing signal emission (Figure 1). For DEIA-WZ021, recombinant RBD protein is immobilized on the plate instead of human ACE2 and HRP labelled recombinant human ACE2 is added into the plate to competitive with neutralizing antibody (Figure 2). This assay format allows for the detection of all neutralizing antibodies independent of isotype, including IgA, which is particularly salient to mucosal tissue infections.
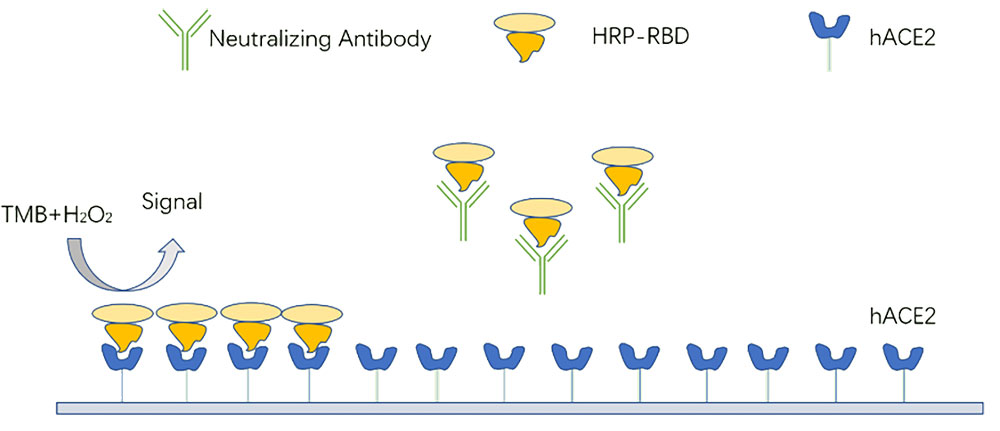 Figure 1. Assay Principle of Creative Diagnostics SARS-CoV-2 Neutralizing Antibody ELISA Kit (CD Cat# DEIASL055)
Figure 1. Assay Principle of Creative Diagnostics SARS-CoV-2 Neutralizing Antibody ELISA Kit (CD Cat# DEIASL055)
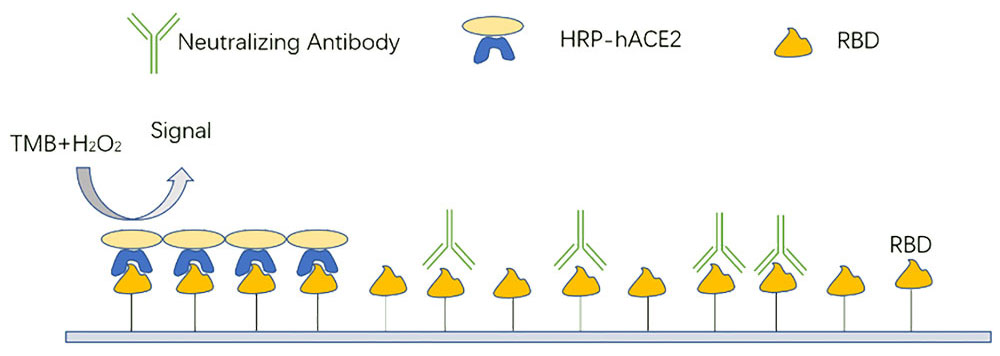 Figure 2. Assay Principle of Creative Diagnostics SARS-CoV-2 Neutralizing Antibody ELISA Kit (CD Cat# DEIA-WZ021)
Figure 2. Assay Principle of Creative Diagnostics SARS-CoV-2 Neutralizing Antibody ELISA Kit (CD Cat# DEIA-WZ021)
Since the outbreak of the new coronavirus pneumonia (Corona Virus Disease 2019, COVID-19), it has rapidly swept across most countries around the world and is still spreading. To deal with the epidemic, it is very urgent to successfully develop a safe and effective vaccine. According to the data released by the WHO a few days ago, there are 198 candidate new coronavirus vaccines being developed and registered in various countries, of which 44 have entered the clinical trial stage, and 10 have entered the phase III clinical trial. In the process of vaccine development and marketing, the presence of neutralizing antibodies can indicate an ability to suppress progression of SARS-CoV-2 infection and is important in evaluating the lifetime and efficacy of specific antibodies in the host.
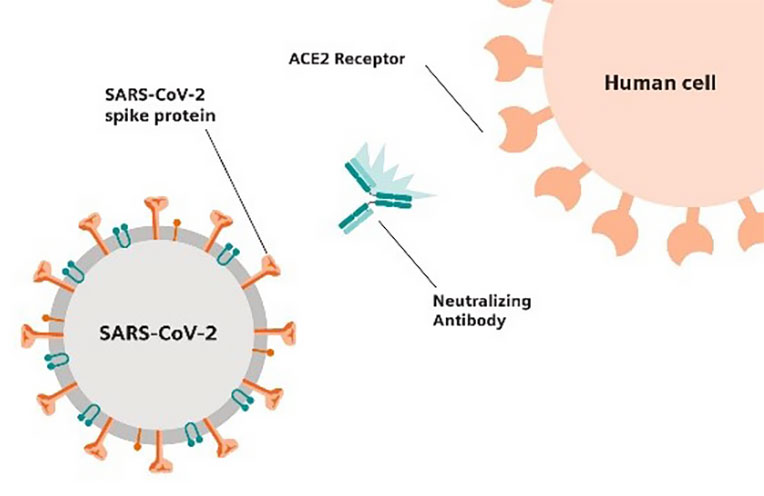 Figure 3. Neutralizing Antibody blocking S protein
Figure 3. Neutralizing Antibody blocking S protein
from binding to ACE2 Receptor.
The SARS-CoV-2 surface glycoprotein, also known as the spike glycoprotein, is located on the outer envelope of the virion and contains the receptor binding domain (RBD), which binds to angiotensin-converting enzyme 2 (ACE2), the functional receptor for SARS-CoV-2. SARS-CoV-2 infection can result in the production of neutralizing antibodies, which bind to the SARS-CoV-2 spike RBD preventing further viral entry and infection, starting approximately 4-10 days after symptom onset (Figure 3). The detection of neutralizing antibodies can provide valuable information for researchers studying immune responses or human immunity to SARS-CoV-2, as well as for the development of effective vaccines.
| Cat. No. | Product Name | Coating | |
| DEIASL055 | SARS-CoV-2 Neutralizing Antibody Assay Kit | hACE2 | Inquiry |
| DEIA-WZ021 | SARS-CoV-2 Neutralizing Antibody Assay Kit | RBD | Inquiry |
| Recombinant Human ACE2 | ||
| DAGC152 | Recombinant Human ACE2 Protein [Fc] (HEK293) | Inquiry |
| DAGC226 | Recombinant Human ACE2 Protein [His] (HEK293) | Inquiry |
| Recombinant SARS-CoV-2 Spike Protein | ||
| DAGC149 | Recombinant SARS-CoV-2 S1 RBD [His] (HEK293) | Inquiry |
| DAGC173 | Recombinant SARS-CoV-2 S1 RBD [hFc] (HEK293) | Inquiry |
| DAGC174 | Recombinant SARS-CoV-2 S1 RBD [mFc] (HEK293) | Inquiry |
| Humanized SARS-CoV-2 Neutralizing Antibody | ||
| CABT-CS033 | SARS-CoV-2 S1 Monoclonal antibody, clone BIB114 (Humanized) | Inquiry |