Immune checkpoints are regulators of the immune system. These pathways are crucial for self-tolerance, which prevents the immune system from attacking cells indiscriminately. Therefore, an immune response is tightly regulated, and multiple mechanisms are in place to prevent autoimmune reactions to self-proteins. These mechanisms are implemented by immune checkpoint molecules.Checkpoint molecules include cytotoxic T lymphocyte antigen-4, programmed death-1, lymphocyte activation gene-3, T-cell immunoglobulin and mucin protein-3, and several others. The expression of these checkpoint molecules on T cells represents an important mechanism that the immune system uses to regulate responses to self-proteins. Recent clinical data show that these Checkpoint molecules play a critical role in objective tumor responses and improved overall survival.
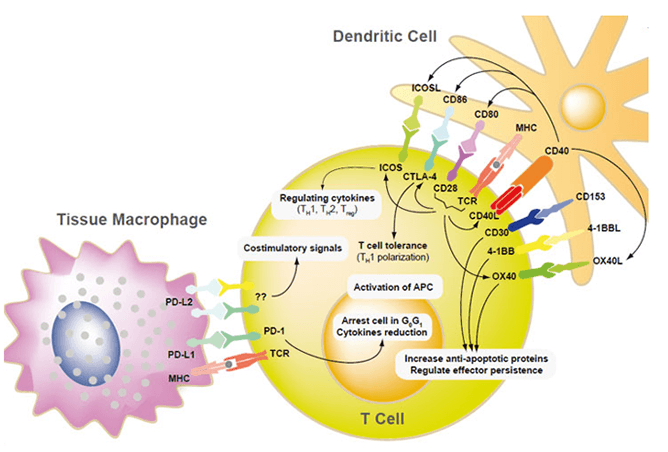 Figure 1. Selected Immune Checkpoint Pathways.
Figure 1. Selected Immune Checkpoint Pathways.
Adopted from P.Sharma 2012 ASCO Annual Meeting.
There are two types of checkpoint molecules in the immune checkpoint signaling pathway. One is to activate the immune system and the other is to suppress the immune system. They are cell surface receptors that cause changes in cell function by binding to ligands secreted by other cells. The researchers completed the classification of these immune checkpoint molecules through the detection of animal defect model phenotypes.
Stimulatory checkpoint molecules
There are eight activation checkpoint molecules. Four stimulatory checkpoint molecules are members of the tumor necrosis factor (TNF) receptor superfamily - CD27, CD40, OX40, GITR and CD137. Another two stimulatory checkpoint molecules belongs to the B7-CD28 superfamily - CD28 itself and ICOS.
Inhibitory checkpoint molecules
Due to the superior performance of immune checkpoint inhibitors in the treatment of tumors, they have received more attention and research from scholars.
Activation
After the activation immune checkpoint molecule binds to its corresponding ligand, it sends an activation signal to the T cell. The T cell is moderately activated. Activation of CD4+ T cells occurs through the simultaneous engagement of the T-cell receptor and a co-stimulatory molecule (like CD28, or ICOS) on the T cell by the major histocompatibility complex (MHCII) peptide and co-stimulatory molecules on the APC. Both are required for production of an effective immune response. The signaling pathways downstream from co-stimulatory molecules usually engages the PI3K pathway generating PIP3 at the plasma membrane and recruiting PH domain containing signaling molecules like PDK1 that are essential for the activation of PKCθ, and eventual IL-2 production, Which in turn activates the entire immune system.
Suppressor
Under normal conditions, ligands that bind to inhibitory immune checkpoint molecules are secreted so that the immune system can be immunized against self-proteins. Some of these pathways have already been described, and some are still unclear. In the proved pathway, we know CTLA-4 is a homolog of CD28 and plays an important role in the development of peripheral tolerance to self-proteins. B7–1 (CD80) and B7–2 (CD86) are the main ligands for CTLA-4. Upon binding B7-1 or B7-2, CTLA-4 recruits the phosphatases SHP2 and PP2A via the YVKM motif in its cytoplasmic domain. SHP2 recruitment results in attenuation of TCR signaling by dephosphorylating the CD3ζ chain. PP2A recruitment results in downstream dephosphorylation of AKT, further dampening the T cell activation pathway. PD-1 ligation by PD-L1 or PD-L2 also recruits SHP2 to the ITSM domain, resulting in membrane proximal decreases in TCR signaling. LAG-3 signaling is dependent on interaction with its ligand, MHC II, as well as its intracellular KIEELE domain. TIM-3 binds to Galectin-9, as well as other ligands. In the absence of ligand binding, TIM-3 is associated with Bat3, protecting the cell from TIM-3–mediated inhibition and allowing for greater activation. However, once TIM-3 binds to Galectin-9, Y265 is phosphorylated and the interaction with Bat3 is disrupted, allowing TIM-3 to deliver inhibitory signals to the T cell. BTLA and CD160 bind to herpes virus entry mediator. BTLA contains an intracellular ITIM domain that may be important in signaling. 2B4 has four intracellular ITSM domains and binds to CD48, but further signaling mechanisms are poorly understood.
Cancer Immunotherapy
The first immune checkpoint receptor, cytotoxic T lymphocyte antigen 4 (CTLA-4), which was discovered and identified, first showed a close relationship with cancer treatment. In animal models, antibodies that block CTLA-4 can mediate the regression of established tumors. This observation directly resulted in the clinical of detection of CTLA-4 monoclonal antibodies in cancer patients. The main limitation of CTLA-4 blocking therapy is the high rate of toxic effects of the immune. Many of the adverse reactions that are present in patients treated with anti-CTLA-4, such as rash, colitis, thyroiditis, and hepatitis. Dozens of immune checkpoint molecules, the next direct clinical immune checkpoint is the inclusion of a programmed death molecule 1. It is an inhibitory receptor expressed on activated tumor-specific CD4-positive cells and CD8-positive T-lymphocytes, and its primary ligand, PD-L1, is also expressed in various types of cancer. Unlike early activated CTLA-4, which modulates systemic T lymphocyte immunity, the PD-1 checkpoint only regulates the activity of cytotoxic T lymphocyte migration into tumors. The expression of PD-L1 ligand is selective and is not over-expressed in normal inflammatory tissues. This makes it possible to block the biological effects of the PD-1 pathway drug, which is much less toxic than anti-CTLA-4. The initial success of cancer immunotherapy by directly activating anti-tumor immunity with tumor vaccines or recombinant cytokines or by injecting prepared tumor-specific immune cells has led to limited success. Until scientific research can better understand the interaction between cancer and the immune system, tumor immunology will take a huge leap forward.