G-protein-coupled receptors (GPCRs), also known as seven-(pass)-transmembrane domain receptors, are the largest family and most diverse group of membrane receptors in eukaryotes. In addition to cell surface, GPCRs are suggested to distribute in intracellular compounds including the endoplasmic reticulum, Golgi apparatus, nuclear membrane and even inside the nucleus itself. There are hundreds of different GPCR proteins, which can bind to diverse set of ligands, including peptide hormones, neurotransmitters, neuropeptides, biogenic amines, amino acids, ions, chemokines, lipid-derived mediators, proteases peptides, proteins, photons and odor molecules. Furthermore, there are orphan GPCRs whose endogenous ligand has not been identified. GRCPs, therefore, are involved in a wide variety of physiological and pathological processes, such as regulation of immune system activity and inflammation, autonomic nervous system transmission, behavioral and mood regulation, sensory transmission, homeostasis modulation and even growth and metastasis of several types of tumors. According to the pharmacological properties, GPCRs can be divided into four catalogs: the largest rhodopsin family, the secretion and adhesion family, the metabotropic glutamate family, and the frizzled family.
The main feature of GPCR structure is the seven transmembrane (7TM) -spanning segments connected by three intracellular (EL1-EL3) and three extracellular (IL1-IL3) loops. Members of the GPCR superfamily share the same basic architecture: 7TM α-helices, an extracellular amino-terminal segment and an intracellular amphipathic helix and the carboxy-terminal tail.
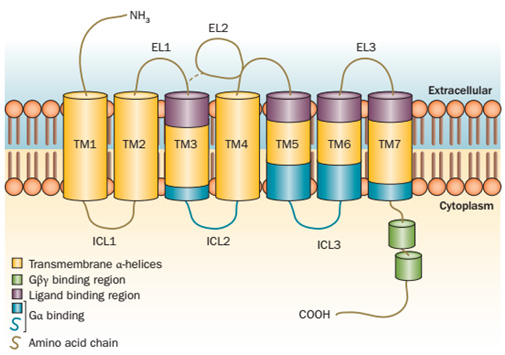 Figure 1. Schematic diagram of GPCR structure. (Elena N. et al., 2014)
Figure 1. Schematic diagram of GPCR structure. (Elena N. et al., 2014)
GPCRs, as their name implies, interact with G proteins in the plasma membrane. The signaling cascades of a GPCR initiate from the binding of an external signaling molecule in the form of a ligand or other signal mediator. This causes a conformational change in the receptor and triggers the interaction between GPCR and a nearby G protein, leading to the activation of a G protein. This process is suggested to be regulated by a guanine nucleotide-binding complex (consisting of Gα, Gβ, and Gγ subunits), as well as additional regulatory proteins. In inactive state, the GPCR is bound to a heterotrimeric G protein complex, the Gα subunit is bound to GDP and is firmly attached to the Gβ and Gγ subunits. Binding of a ligand to the GPCR results in a conformational change of the receptor, which leads to the transmission of bound Gα subunit to active status, along with the exchange of GDP to GTP as well as the dissociation of Gα subunit from the Gβγ dimer and from the receptor.
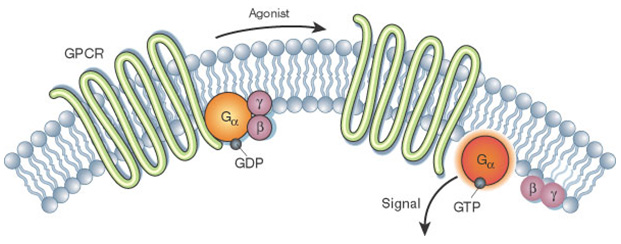 Figure 2. Activation of the G alpha subunit of a GPCR. (Li, J. et al., 2002)
Figure 2. Activation of the G alpha subunit of a GPCR. (Li, J. et al., 2002)
The signal is terminated by the hydrolysis reaction of GTP, which restores another heterotrimeric G protein to reform new G-protein binding complex that is ready to initiate another round of signal transduction. However, the dissociated Gα and Gβγ subunits will interact with other intracellular proteins to continue the signal transduction cascade.
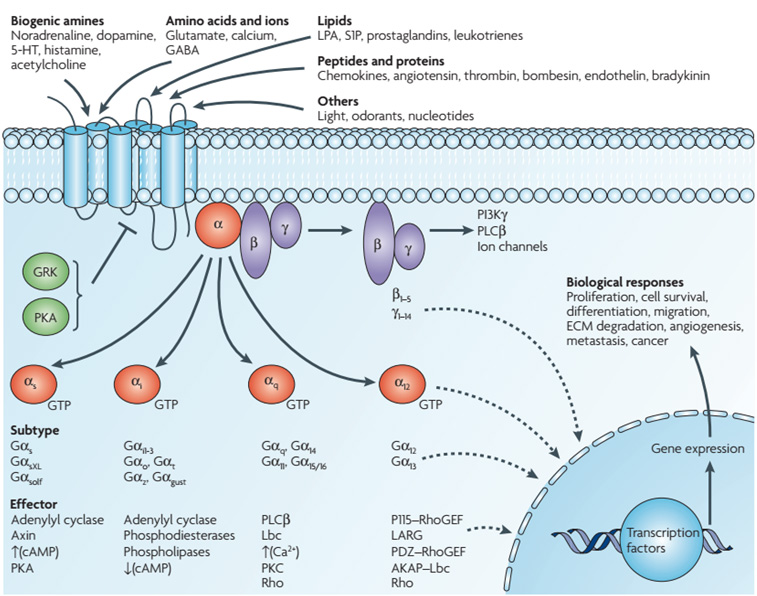 Figure 3. Diversity of G-protein-coupled receptor signaling. (Robert D. et al., 2007)
Figure 3. Diversity of G-protein-coupled receptor signaling. (Robert D. et al., 2007)
Typically, Gαs stimulates adenylyl cyclase (AC) and increases cyclic AMP (cAMP) levels, whereas Gαi inhibits AC and decreases cAMP levels. Gαq family members activate phospholipase C (PLC), which cleaves phosphatidylinositol bisphosphate (PIP2) into diacylglycerol and inositol triphosphate (IP3), and mediates the activation of PKC and CaMKII. The Gβ and Gγ subunits function as a dimer to activate numerous signaling molecules (e.g. phospholipases, ion channels and lipid kinases). In addition to the modulation of these classical second-messenger generating systems, Gα subunits and Gβγ subunits are reported to regulate the activation of key intracellular signal-transducing molecules, such as small GTP-binding proteins of the Ras and Rho families and members of the mitogen-activated protein kinase (MAPK) family of serine-threonine kinases, including extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK) and p38. Src family kinases are recruited following activation of PI3Kγ by β/γ subunits. Ultimately, the integration of the functional activity of the G-protein-regulated signaling networks control normal cellular functions mentioned above, otherwise the aberrant activity of G proteins and their downstream target molecules may be involved in several pathological conditions including cancer progression and metastasis.