CFTR is a chloride channel located on the cell membrane. Under the mediation of cAMP, CFTR is phosphorylated, causing the channel to open and transporting about 10 CIs extracellularly per minute. When the cftr gene is mutated (most commonly, the codon encoding 508 phenylalanine is lost), the defective CFTR cannot be processed normally in the endoplasmic reticulum, and most cannot be transported to the cell membrane; even a small number of mutant CFTR proteins can be transported to the cell membrane, but due to structural abnormalities, the function of CFTR ion channel protein is lost, resulting in the inability of epithelial cells to transport CI-, and CI- will accumulate in the cells.
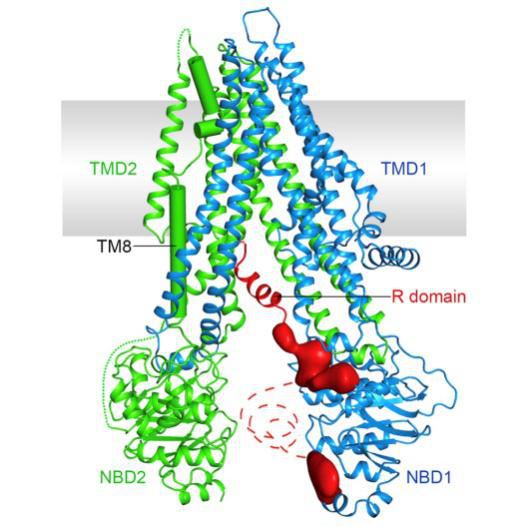
In 1989, Riordan et al first cloned the cftr gene, and in 1991, CFTR was identified as a chloride-selective channel, thus beginning a systematic study of CFTR as a chloride channel. The CFTR gene is in the long arm of human chromosome 7, full-length 250,000 bp, containing 27 exons. The final length of the mature mRNA is 6129 bases, of which 4443 bases can encode the sequence, and the mature CFTR protein is 1480 amino acids, and the relative molecular mass is about 168173. In addition, CFTR is successfully cloned in other organisms, the mouse is located on chromosome 6, and the rat is located on 5 chromosomes. CFTR is widely distributed and is expressed in many cell membranes such as lung, liver, pancreas, intestine, and gonads. Although it is called chloride channel, it also involves the transport of other ions. Since the most important physiological action is the transport of chloride ions, it is called a chloride ion channel. CFTR is a transmembrane protein, which is difficult to obtain ideal crystals. So far, no complete structural images have been obtained, but since it belongs to the ABC family P-glycoprotein, the structural similarity indicates the rationality of the speculation. It is now certain that CFTR consists of five functional domains: two transmembrane domains, MSD1 and MSD2; two nucleotide binding domains, NBD1 and NBD2, in which two MSDs form selective chloride channels. While the two NBD domains regulate the gating of the chloride ion by phosphorylation the r gene.
CFTR is a chloride channel protein that transports chloride ions from the cell to the outside of the cell. Due to its important physiological role, it is often used clinically as a target for the treatment of diseases. Autosomal dominant polycystic kidney disease is a hereditary disease characterized by multiple intravesicular vesicles. Lesions damage like kidney function, can lead to secondary hypertension and renal failure. There is currently no cure for polycystic kidney therapy. Kidney dialysis or kidney transplantation is required in the later stages of the disease. The detailed mechanism of vesicle formation in the kidney of patients with polycystic kidney disease is currently unclear, but the growth of vesicles involves the proliferation of vesicle epithelial cells and the secretion of fluid in vesicles. Previous studies have suggested that fluid secretion in vesicles is caused by the passive secretion of water molecules by the chloride channel CFTR actively secreting chloride ions. Liquid secretion causes an increase in pressure within the vesicle and excessive growth of the vesicle. Therefore, inhibition of the chloride channel CFTR has the potential to inhibit vesicle growth by reducing chloride ion secretion. Studies using cell, organ and whole model studies have found that the CFTR inhibitors tetraozl-172 have a significant inhibitory effect on the occurrence and development of vesiclesin vitro and in vivo.The inhibitor of CFTR significantly inhibited the formation and rapid growth of vesicles in Matrigel induced by forskolin and did not exhibit cytotoxicity.In vitroculture of embryonic kidney model also showed that CFTR inhibitor significantly inhibited cAMP-induced vesicle growth in a dose-response relationship. Subcutaneous injection of CFTR inhibitors in polycystic kidney mice produces a stable therapeutic effect. Injection of CFTR inhibitor for 7 days significantly inhibited the formation and growth of renal vesicles in the kidney and relieved renal damage caused by polycystic kidney disease. The experimental results suggest that CFTR inhibitors can inhibit the growth of polycystic kidney vesicle fluid by inhibiting its secretion. The chloride channel CFTR can be used as a new target for polycystic kidney therapy. The CFTR protein also regulates the function of other channels, such as transporting sodium ions on the cell membrane. These ion channels are essential for maintaining the function and tissue homeostasis of the lung and other organ epithelial cells. Studies have shown that functional defects or deletions caused by mutations in the cftrgene are responsible for cystic fibrosis in the lungs. In addition, more and more studies have shown that the cftr gene is closely related to many tumorigeneses and plays a role in promoting or inhibiting tumorigenesis in different tumorigenesis. Recent studies have confirmed that CFTR is a tumor suppressor gene for human intestinal cancer. Inhibition of CFTR expression promotes epithelial-mesenchymal transition (EMT) characteristics of breast cancer cells; whereas overexpression of CFTR inhibits proliferation and migration of endometrial tumor cells. These results indicate that CFTR has the function of a tumor suppressor gene in the above tumor. However, in prostate cancer and nasopharyngeal carcinoma (NPC), cftr gene functions as a proto-oncogene. These results clearly indicate that cftrgene expression is closely related to malignant changes and drug resistance of tumor but may play different roles and mechanisms in different types of tumors.
Reference: