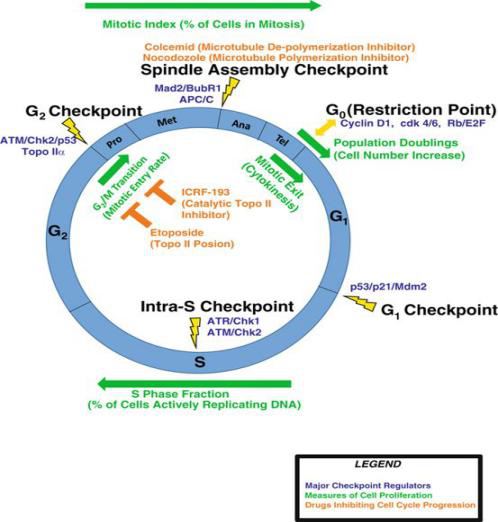
The cell cycle refers to the whole process that the cell undergoes from the completion of one division to the end of the next division, and is divided into two phases, the interval, and the division phase. Life is a continuous process that passes from one generation to the next, so it is a process of constantly updated and constantly starting from scratch. The life of a cell begins with the division of its mother cell, the formation of its daughter cells, or the death of the cell itself. The formation of daughter cells is usually a sign of the end of a cell division, which refers to the process that occurs from the time when a cell divides to form a daughter cell until the next cell divides to form a daughter cell. In this process, the genetic material of the cell is replicated and equally distributed to the two daughter cells. The precise regulation of the cell cycle requires the participation of many factors. Cell cycle disorders can lead to many important diseases, such as cancer, cardiac hypertrophy and anemia. Therefore, further research on the cell cycle is particularly important.
The cell cycle can be divided into interval, pre-, mid-, post-, and end phase. The interval is further divided into three phases, namely, the pre-DNA synthesis phase (G1 phase), the DNA synthesis phase (S phase), and the late DNA synthesis phase (G2 phase). The pre-chromatin filaments are highly helical and gradually form chromosomes. The chromosome is short and thick, and strongly basophilic. The two centrosomes move in opposite directions to form two poles in the cell; then the microtubules are synthesized starting from the central granule with the body as a starting point to form a spindle. As the nucleolus phase spirals with the chromatin, the nucleolus gradually disappears. The nuclear envelope begins to collapse into a discrete vesicular endoplasmic reticulum. The metaphase cells become spherical, and the nucleolus and nuclear envelope have completely disappeared. The chromosomes are all moved to the equatorial plane of the cell, and the microtubules emanating from the poles of the spindle are attached to the centromere of each chromosome. From the metaphase cells, a complete chromosome group can be isolated, a total of 46, of which 44 are autosomes and 2 are sex chromosomes. The male karyotype is 44+XY and the female is 44+XX. The isolated chromosomes are short, thick or hairpin-like, and are composed of two chromatids linked by narrow centromere. In the anaphase, due to the activity of the spindle microtubules, the centromere is longitudinally split, and the two chromatids of each chromosome are separated and moved in opposite directions, approaching the respective centrosomes, and the chromatids are divided into two groups. At the same time, the cells are elongated, and due to the activity of the microfilament bundle under the cell membrane of the equator, the part is narrowed, and the cells are dumbbell-shaped. The telophase chromatid gradually de-screws, and re-exists chromatin filaments and nucleoli; endoplasmic reticulum vesicles are combined into nuclear envelope; group equator is narrowed and deepened, and finally splits into two diploid daughter cells.
There are two stages in the cell cycle that are most important, G1 to S and G2 to M; these two stages are in the period of complex and active molecular level changes, which are easily affected by environmental conditions. If they can be artificially regulated, they will be important to understand the growth and development of organisms and control tumor growth. Manyin vivo factors have been found to stimulate or inhibit cell proliferation, such as multiple hormones, serum factors, polyamines, proteolytic enzymes, neuraminidase, cAMP, cGMP, diglycerides (DG), inositol triphosphate (IP3) and more. Increased intracellular cAMP concentration has an inhibitory effect on cell proliferation. Any factor that can increase intracellular cAMP can inhibit cell proliferation and decrease cell growth rate. Conversely, any factor that can decrease intracellular cAMP content can promote DNA synthesis and proliferation of cells. There are also many experiments that indicate that cGMP also regulates cell proliferation. For example, when cGMP or dibutyryl cGMP is added to 3T3 cells resting in G1 phase, it can induce an increase in DNA synthesis and promote cell division. For instance, increased cell cGMP levels can promote cell mitosis, in turn, drugs that promote mitosis can also increase the concentration of cGMP. cAMP can inhibit cell division and promote cell differentiation. cGMP can inhibit cell differentiation and promote cell proliferation. In normal growth cells, cAMP and cGMP are maintained at appropriate levels to regulate cell cycle. Inhibin is a small molecule protein or polypeptide produced by cells, and some also contain sugar or RNA. It is not species-specific, but has cell specificity, inhibits the proliferation of similar cells and is reversible. When the content of the inhibitor reaches a certain concentration, the proliferation of the same type of cells can be inhibited, and when the concentration of inhibin is decreased, the proliferation of cells is active. Some people think that the mechanism of the action of inhibitor is that it can activate the adenylyl cyclase activity on the cell membrane, increase the concentration of intracellular cAMP, thus inhibit the proliferation of cells, and may also phosphorylate proteins through cAMP-dependent protein kinase to influence the activity of regulating genes.
Reference: