Bone morphogenetic proteins ( BMPs ) are a group of growth factors also known as cytokines and as metabologens. BMPs are secreted signaling molecules that belong to the transforming growth factor β family of growth and differentiation factors. Bone morphogenetic proteins are synthesized as large precursors consisting of a signal peptide, a prodomain and a carboxy terminal region of 100 to 125 amino acids. Within the carboxy terminal region of most bone morphogenetic ptoteins are seven highly conserved cysteine residues important for correct folding of these dimeric molecules.Bone morphogenetic protein receptors are members of a larger family of serine threonine kinases that include the receptors for transforming growth factor vetas, activins and inhibins.
Originally, seven such proteins were discovered. Of these, six (BMP2 through BMP7) belong to the transforming growth factor beta superfamily of proteins. BMP1 is a metalloprotease. Since then, thirteen more BMPs have been discovered, bringing the total to twenty.
Table 1. BMP family related products
Bone morphogenetic protein 1, also known as BMP1, is a protein which in humans is encoded by the BMP1 gene. BMP-1 is implicated in de novo endochon- dral bone formation and is also a met- alloprotease with multiple sites for potential N-linked glycosylation and EGF-like se- quences that may confer calcium depen- dence on binding activities of adjacent CUB domains.
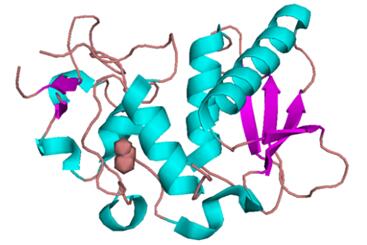 Figure 1. Crystal Structure of BMP1 Protease Domain.
Figure 1. Crystal Structure of BMP1 Protease Domain.
Bone morphogenetic protein 2 or BMP-2 belongs to the TGF-β superfamily of proteins. Like many other proteins from the BMP family, BMP-2 has been demonstrated to potently induce osteoblast differentiation in a variety of cell types. It is involved in the hedgehog pathway, TGF beta signaling pathway, and in cytokine-cytokine receptor interaction. It is also involved in cardiac cell differentiation and epithelial to mesenchymal transition.
Bone morphogenetic protein 3, also known as osteogenin, is a protein in humans that is encoded by the BMP3 gene. The protein encoded by this gene is a member of the transforming growth factor beta superfamily. It, like other bone morphogenetic proteins (BMP's) is known for its ability to induce bone and cartilage development. It is a disulfide-linked homodimer. It negatively regulates bone density. BMP3 is an antagonist to other BMP's in the differentiation of osteogenic progenitors. It is highly expressed in fractured tissues.
BMP4 is found on chromosome 14q22-q23. Yielding an active carboxy-terminal peptide of 116 residues, human bmp4 is initially synthesized as a forty percent residue preproprotein which is cleaved post translationally. BMP4 has seven residues which are conserved and glycosylated. The monomers are held with disulphide bridges and 3 pairs of cysteine amino acids. This conformation is called a “cystine knot”. BMP4 can form homodimers or heterodimers with similar BMPS.
BMP5 may play a role in certain cancers. Like other BMP's BMP5 is inhibited by chordin and noggin. It is expressed in the trabecular meshwork and optic nerve head and may have a role in the development and normal function. It is also expressed in the lung and liver.
BMP6 is able to induce all osteogenic markers in mesenchymal stem cells. BMPs were originally identified by an ability of demineralized bone extract to induce endochondral osteogenesis in vivo in an extraskeletal site. Based on its expression early in embryogenesis, the BMP encoded by this gene has a proposed role in early development. In addition, the fact that this BMP is closely related to BMP5 and BMP7 has led to speculation of possible bone inductive activity.
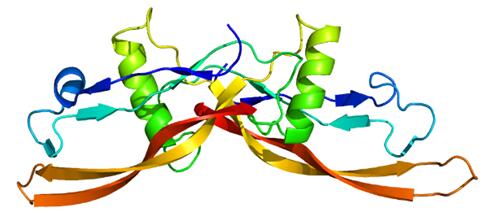 Figure 2. Structure of the BMP6 protein.
Figure 2. Structure of the BMP6 protein.
Bone morphogenetic protein 7 or BMP7 (also known as osteogenic protein-1 or OP-1) is a protein that in humans is encoded by the BMP7 gene. It plays a key role in the transformation of mesenchymal cells into bone and cartilage. It is inhibited by noggin and a similar protein, chordin, which are expressed in the Spemann-Mangold Organizer. BMP7 may be involved in bone homeostasis. It is expressed in the brain, kidneys and bladder. BMP7 induces the phosphorylation of SMAD1 and SMAD5, which in turn induce transcription of numerous osteogenic genes. It has been demonstrated that BMP7 treatment is sufficient to induce all of the genetic markers of osteoblast differentiation in many cell types.
BMP8A may be involved in epithelial osteogenesis. It also plays a role in bone homeostasis. It is a disulfide-linked homodimer. Bone morphogenetic protein 8B is a protein that in humans is encoded by the BMP8B gene. It has close sequence homology to BMP7 and BMP5 and is believed to play a role in bone and cartilage development. It has been shown to be expressed in the hippocampus of murine embryos.
BMP10 is a polypeptide belonging to the TGF-β superfamily of proteins. It is a novel protein that, unlike most other BMP's, is likely to be involved in the trabeculation of the heart. Bone morphogenetic proteins are known for their ability to induce bone and cartilage development. BMP10 is categorized as a BMP since it shares a large sequence homology with other BMP's in the TGF-β superfamily.
BMP11 has been shown to suppress neurogenesis through a pathway similar to that of myostatin, including stopping the progenitor cell-cycle during G-phase. The similarities between BMP11 and myostatin imply a likelihood that the same regulatory mechanisms are used to control tissue size during both muscular and neural development. In the mouse adult central nervous system, BMP11 alone can improve the cerebral vasculature and enhance neurogenesis.
Bone morphogenetic protein 15 is a protein that in humans is encoded by the BMP15 gene. It's mainly involved in folliculogenesis. It is a paracrine signaling molecule involved in oocyte and follicular development. Using Northern blot analysis, BMP15 has been shown to be exclusively expressed in the ovaries. It is thought that this protein may be involved in oocyte maturation and follicular development as a homodimer or by forming heterodimers with a related protein, Gdf9.
Recombinant BMP when implanted with an appropriate collagenous matrix is capable of inducing new bone at ectopic sites, and this effect is dependent on the dose. As a member of the transforming growth factor-beta superfamily, BMP signaling regulates a variety of embryonic patterning during fetal and embryonic development. For example, BMP signaling controls the early formation of the mullerian duct (MD) which is a tubular structure in early embryonic developmental stage and eventually becomes female reproductive tracts. Chemical inhibiting BMP signals in chicken embryo cause a disruption of MD invagination and block the epithelial thickening of the MD-forming region, indicating that the BMP signals play a role in early MD development.
Disruption of BMP signaling can affect the body plan of the developing embryo. For example, BMP4 and its inhibitors noggin and chordin help regulate polarity of the embryo. Specifically BMP-4 and its inhibitors play a major role in neurulation and the development of the neural plate. BMP-4 signals ectoderm cells to develop into skin cells, but the secretion of inhibitors by the underlying mesoderm blocks the action of BMP-4 to allow the ectoderm to continue on its normal course of neural cell development.
Moreover, BMP signaling is involved in the formation of foregut and hindgut, intestinal villus patterning, and endocardial differentiation. Villi contribute to increase the effective absorption of nutrients by extending the surface area in small intestine. Gain or lose function of BMP signaling altered the patterning of clusters and emergence of villi in mouse intestinal model. BMP signal derived from myocardium is also involved in endocardial differentiation during heart development. Inhibited BMP signal in zebrafish embryonic model caused strong reduction of endocardial differentiation, but only had little effect in myocardial development.
The BMP ligands, receptors and antagonists extracellularly are capable of impacting tumor growth and progression, both positively and negatively. Depending on the tumor cell type (carcinoma versus sarcoma) and stage (primary versus metastasis), BMPs can affect cancer growth and its progression and modulate responsiveness to endocrine and metabolic factors.
Mutations in BMPs and their inhibitors are associated with a number of human disorders which affect the skeleton.
Bone morphogenetic protein 2 is shown to stimulate the production of bone. Recombinant human protein (rhBMP-2) is currently available for orthopaedic usage in the United States. The use of dual tapered threaded fusion cages and recombinant human bone morphogenetic protein-2 on an absorbable collagen sponge obtains and maintains intervertebral spinal fusion, improves clinical outcomes, and reduces pain after anterior lumbar interbody arthrodesis in patients with degenerative lumbar disc disease.
BMP3 is hypermethylated in many cases of colorectal cancer (CRC) and hence along with other hypermethylated genes, may be used as an biomarker to detect early stage CRC.
Increase in expression of BMP4 has been associated with a variety of bone diseases, including the heritable disorder Fibrodysplasia Ossificans Progressiva. There is strong evidence from sequencing studies of candidate genes involved in clefting that mutations in the bone morphogenetic protein 4 (BMP4) gene may be associated in the pathogenesis of cleft lip and palate.
Human recombinant BMP7 can be used to aid in the fusion of vertebral bodies to prevent neurologic trauma. BMP7 also has the potential for treatment of chronic kidney disease. Kidney disease is characterized by derangement of the tubular architecture by both myofibroblast buildup and monocyte infiltration. Because endogenous BMP-7 is an inhibitor of the TGF-β signaling cascade that induces fibrosis, the use of exogenous recombinant BMP-7 (rhBMP-7) could be a viable treatment of chronic kidney disease.
Defects in BMP15 are associated with primary ovarian insufficiency. BMP15 may represent a biomarker of ovarian response to ovarian stimulation or oocyte quality.
References: