Creative Diagnostics understand that each customer has unique requirements for their project. That's why we are completely flexible with custom purification for list antibody products. We can custom provide 0.2 micron sterile-filtered, Low Endotoxin and Azide-Free antibodies for in vivo and in vitro assays. We are also committed to provide the highest quality and standards.
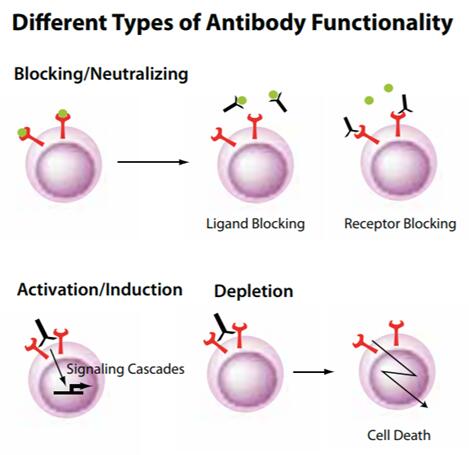
Functional antibodies can either mimic or interrupt the natural biologic effects associated with ligand-receptor interactions, or have a physiological effect on a target cell or molecule. They may be used in several functional assays, including depletion, activation, neutralization, or blocking experiments, both in vitro and in vivo. Functional grade antibodies are available free of preservatives and tested for low endotoxin content.
Endotoxins, toxic substance bound to the bacterial cell wall and released when the bacterium ruptures or disintegrates. Removal of endotoxin is one of the most difficult downstream processes during antibody purification, because endotoxin is extremely heat and pH stable. Our team has developed a low endotoxin purification process that serves to minimize endotoxin loads. Besides, our rapid test methods allow us to produce results right at the point of antibody purification.
Sodium azide is a preservative used for inhibiting the growth of contaminants, such as bacteria or fungi. However, its presence in antibody solutions can affect the use of the antibody in cell culture assays as it is toxic to cells. It can also interfere with antibody conjugation and inhibits the activity of the enzyme horseradish peroxidase. Many CD antibody products contain sodium azide and this information is provided on individual datasheets. If the antibody is to be used for cell culture assays or conjugation, sodium azide removal from the antibody solution is recommended. Creative Diagnostics can provide sodium azide removal service to meet customer's needs.
The following three procedures can be used to remove azide: