The execution phase of apoptosis is initiated by the cleavage of Caspase-3 and results in destruction of the nucleus, DNA fragmentation, degradation of cytoskeletal and proteins, chromatin condensation, formation of apoptotic bodies, expression of ligands for phagocytic cell receptors and finally uptake by phagocytic cells.
Apoptosis execution pathway is the same terminal of extrinsic, intrinsic, and perforin/granzyme apoptosis pathways, leading to the morphological and biochemical changes that characterize apoptosis. As the final stage of apoptosis, it causes cell shrinkage, formation of cytoplasmic blebs and apoptotic bodies and finally phagocytosis of the apoptotic bodies by adjacent parenchymal cells, neoplastic cells or macrophages.
Extrinsic apoptosis pathway and intrinsic apoptosis pathway are two major pathways lead to apoptosis execution, as well as a perforin/granzyme apoptosis pathway. The extrinsic apoptosis pathway that initiates apoptosis is triggered by a death ligand binding to a death receptor present in the plasma membrane. Intrinsic apoptosis pathway initiates apoptosis by activating Caspase-3 or by cleaving BH3 interacting domain death agonist (Bid) through the Puma and Noxa protein subgroup. Perforin/granzyme apoptosis pathway is the primary signaling pathway used by cytotoxic lymphocytes (Cytotoxic T cells) to eliminate virus-infected and/or transformed cells. Each apoptosis pathway needs specific triggering signals to begin an energy-dependent cascade of molecular events and activates its initiators Caspase-8/9/10 which in turn activates the executioner Caspase-3 in the execution pathway.
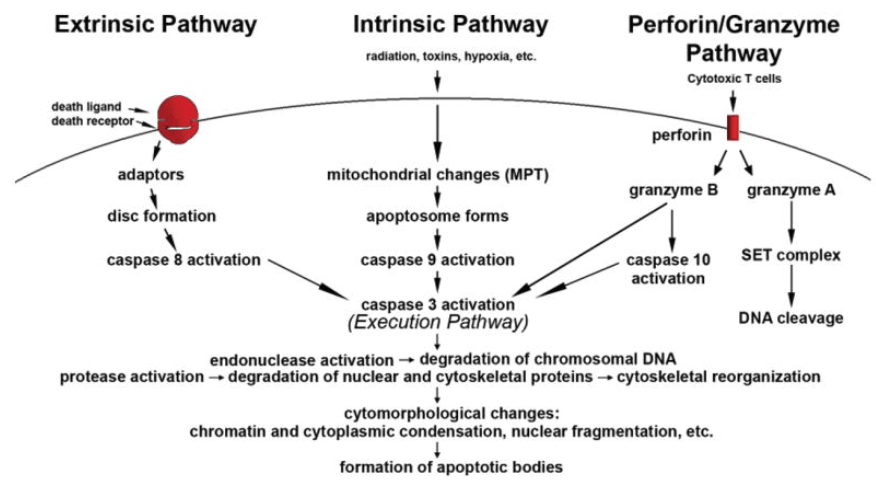 Figure 1. An overview of apoptosis execution pathway. (Elmore, S. 2007)
Figure 1. An overview of apoptosis execution pathway. (Elmore, S. 2007)
Apoptosis execution pathway begins with the activation of the execution caspases which activate cytoplasmic endonuclease, and degrade nuclear material, and protease that degrades the nuclear and cytoskeletal proteins. Caspase-3/6/7 function as effector or “executioner” caspases cleaving various substrates including PARP, cytokeratin, fodrin (the plasma membrane cytoskeletal protein), α-tubulin protein required for the establishment of cell morphology, and the nuclear protein NuMa, which ultimately causes the morphological and biochemical changes seen in apoptotic cells. All the extrinsic, intrinsic and perforin/granzyme apoptosis pathways will end at the point of the execution phase.
The mitochondrial changes (MPT) lead to cytochrome-c and Smac releases from mitochondria followed by apoptosome formation and caspase activation. Specifically, cytochrome-c is complex with APAF-1, recruiting and activating the initiator Caspase-9 which goes on to activate the effector caspases, in particular, Caspase-3 leads to the biochemical and morphological changes characteristic of apoptosis. Caspase activity is inhibited by XIAP, whose action is antagonized through its interactions with the pro-apoptotic Smac. Caspase activation following cytochrome-c and Smac release occurs within minutes, however, this rapid response is significantly slowed when XIAP levels are high which may be particularly important in cancers as XIAP is overexpressed in a number of tumor types.
Caspase-3/6/7 are highly homologous to each other and their final functions are also similar in the apoptosis execution pathway. Caspase-3 is considered to be the most important of the executioner caspases and is activated by any of the initiator Caspase-8/9/10. Caspase-3 activates the endonuclease caspase-activated DNase (CAD) which is complexed with its inhibitor ICAD. In apoptotic cells, activated Caspase-3 cleaves ICAD to release CAD. CAD then degrades chromosomal DNA within the nuclei and causes chromatin condensation. It also induces cytoskeletal reorganization and disintegration of the cell into apoptotic bodies that taken by phagocyte. The phagocytosis of apoptotic cells and fragments is the last component of execution apoptosis. It finally results in disruption of the cytoskeleton, intracellular transport, cell division, and signal transduction.
Apoptosis execution pathway is totally impaired with advanced disease stage in tumor tissue. Patients associated with apoptosis are unable to undergo caspase activation and frequently examined as prognostic indicators in colorectal cancer. It has been shown that APAF-1, Smac, XIAP, and the Caspases-3/9 are differentially expressed between normal tissue and colorectal tumor sand, with increased expression of APAF-1 and Smac and decreased XIAP expression associating with longer patient survival. Report shows that targeting Caspase-3 activation could be a valuable strategy for cancer therapy. However, it is important to note that the decision between apoptosis and survival is not determined solely by individual proteins but rather by the relative abundance and interactions of multiple key apoptotic proteins. Therefore examination of the system as a whole rather than single markers may be more informative for patient prognosis and in identifying targets for therapeutic intervention.
References: