Anaplastic lymphoma kinase (ALK) is an enzyme that is encoded by the ALK gene in humans. Discovered in 1994, it was first identified as a receptor tyrosine kinase in a chromosomal translocation associated with some anaplastic large cell lymphomas(ALCL), where ALK takes its name. ALK was described as a fusion partner, and almost 70~80% of ALK-positive ALCL express the NPM–ALK which is an amino-terminal fusion derived from the nucleophosmin (NPM) protein. NPM itself has multiple functions including the transport of pre-ribosomal particles and ribosome biogenesis, regulation of cell division, DNA repair, transcription and genomic stability, playing a postulated role as either an oncogene or a tumor suppressor depending on its level of expression. NPM-ALK would prove to be the first of many ALK fusion proteins to be described in a wide range of cancer types. In addition to NPM–ALK, other fusions involving ALK include TFG, TPM3 and TPM4, as well as non-nuclear ALK fusion chimeras ATIC, CLTCL1 and moesin.
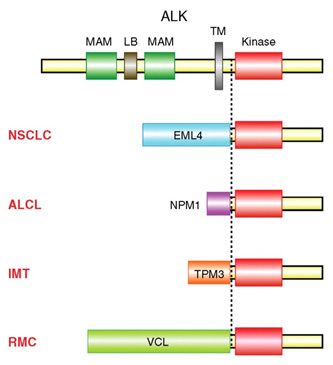 Figure 1. Structural organization of ALK and its fusion proteins. (Mano, H., 2012)
Figure 1. Structural organization of ALK and its fusion proteins. (Mano, H., 2012)
Fusions of ALK have a strong oncogenic potential as the aberrant activity of tyrosine kinase promotes cell survival, cell cycle progression and proliferation, leading to cytoskeletal rearrangements and changes in cell shape. The transformation of oncogenic ALK is mediated by interactions with downstream molecules that trigger substantial intracellular signaling cascades. ALK fusions activate many different pathways, among which the most relevant and characterized pathways are the Ras–extracellular signal-regulated kinase (ERK) pathway, the Janus kinase 3 (JAK3)–STAT3 pathway, the phosphatidylinositol 3-kinase (PI3K)–Akt pathway and the phospholipase C-γ (PLCγ) pathway. These pathways share many interaction points to mediate the effects of ALK activity.
Mitogenic signaling dependent on ALK fusion proteins is mainly resulted from the activation of the Ras–ERK pathway, which is essential mostly for ALCL proliferation. In NPM–ALK, several adaptors or scaffolding molecules with Src-homology 2 (SH2) or phosphotyrosine binding (PTB) domains can bind and activate the Ras–ERK pathway, such as insulin receptor substrate 1 (IRS1), SRC homology 2 domain-containing (SHC) and SRC. They directly bind to the specific tyrosine residues of ALK. Specifically, SHP2–growth factor receptor-bound protein 2 (GRB2) complex is able to bind to different regions of NPM–ALK, and primarily regulates ALK-mediated phosphorylation of SHP2. The interaction of GRB2 with ALK and SHC leads to the enhancement of ERK1/2 phosphorylation through SRC.
PLCγ, docking in residue Y664 of NPM–ALK, is directly activated by ALK, leading to the generation of inositol triphosphate (IP3) and diacylglycerol (DAG). These molecules further modulate the release of calcium stores from the endoplasmic reticulum (ER) and activate protein kinase C (PKC). Interestingly, the PLC-γ docking site (Y664) is present only in the human ALK but not in the mouse gene, suggesting a unique function in human ALK-mediated transformation.
STAT3 is the key mediator involved in the survival mechanisms triggered by ALK fusion proteins. STAT3 is activated by ALK either directly or through JAK3. There are a number of downstream effectors of STAT3, including the B-cell leukemia/lymphoma 2 (BCL2) family of apoptosis-regulating proteins BCL2, BCL-XL and myeloid cell leukemia sequence 1 (MCL1) as well as transcription factor C/EBPβ and surviving. These effectors provide strong anti-apoptotic signaling that inhibits apoptosis and also mediates the spindle assembly checkpoint and cytokinesis. Several studies also demonstrated that ALK–STAT3 may control angiogenesis. In addition to STAT3, STAT5 can also be activated by ALK through JAK2. However, the oncogenic contribution of STAT5 remains poorly defined.
The contribution of PI3K in ALK fusion protein-driven oncogenic process consists of a crucial anti-apoptotic signal. NPM–ALK interacts directly and most likely indirectly with PI3K, which phosphorylates PKB/AKT. AKT can block the function of pro-apoptotic proteins, such as BCL2-antagonist of cell death (BAD), which in turn enhances cell survival. AKT also phosphorylates forkhead box O3A (FOXO3a), leading to the block of FOXO3a-mediated transcription of target genes (such as BIM) that promote apoptosis, cell-cycle arrest and metabolic processes. Finally, the PI3K/AKT pathway also controls the phosphorylation levels of Glycogen synthase kinase (GSK) 3β, upregulating the expression of MCL-1, which may contribute to the neoplastic phenotype.