Adaptor Protein Overview
Adaptor protein plays an important role in cell survival and proliferation modulating signal transduction. Although adaptor proteins themselves do not contain any enzymatic function, their capacity to organize large, temporary protein complexes by linking proteins together in a regulated and selective fashion makes them of outstanding importance in the establishment and maintenance of specificity and efficiency in all known signal transduction pathways. Adaptor proteins contain a variety of protein-binding modules that link protein-binding partners together and facilitate the creation of larger signaling complexes. Adaptor proteins have a variety of functional domains which mediate specific protein-protein and protein-lipid interactions, such as, Src homology 2 (SH2), phosphotyrosine binding (PTB) domains cSrc homology 3 (SH3) domains and PH domains. There are two kinds of them existing in homo cells, one is Signal transducing adaptor proteins (STAPs),another is Vesicular transport adaptor protein.
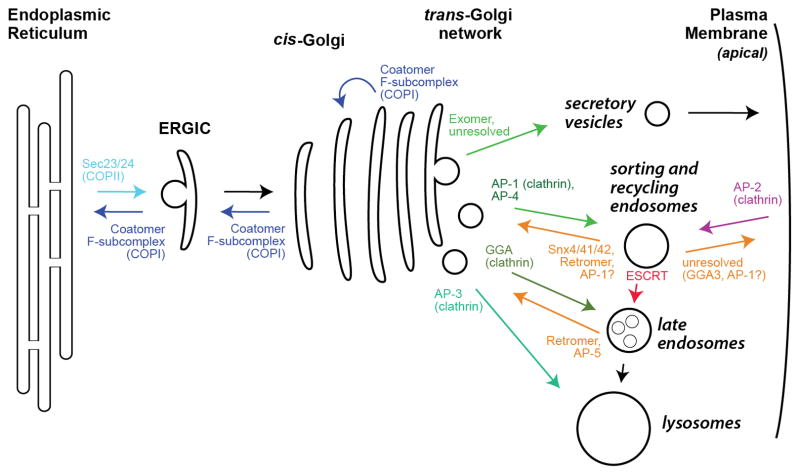
Figure.1 Overview of cargo adaptor localization
Adaptor proteins usually contain several domains within their structure (e.g., Src homology 2 (SH2) and SH3 domains) that allow specific interactions with several other specific proteins. SH2 domains recognise specific amino acid sequences within proteins containing phosphotyrosine residues and SH3 domains recognise proline-rich sequences within specific peptide sequence contexts of proteins. Phosphotyrosine binding (PTB) domains can bind to phosphotyrosine containing motifs. PH domains are lipid interacting modules which bind to specific phosphoinositides and play a role in recruiting proteins to defined regions of the plasma membrane.
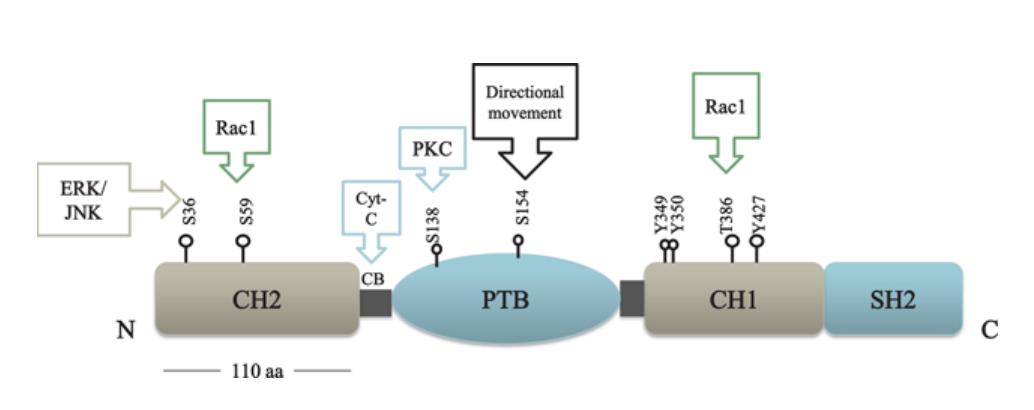
Figure 2. Scheme of adaptor protein structure and examples of interacted substrate
Adaptor Proteins Signal Transduction
Vesicular transport adaptor proteins are proteins involved in forming complexes that function in the trafficking of molecules from one subcellular location to another. These complexes concentrate the correct cargo molecules in vesicles that bud or extrude off of one organelle and travel to another location, where the cargo is delivered. While some of the details of how these adaptor proteins achieve their trafficking specificity has been worked out, there is still much to be learned. There are several human disorders associated with defects in components of these complexes, including Alzheimer's and Parkinson's diseases.
Recent studies have shown that sorting of transmembrane proteins to the basolateral domain of epithelial cells and the somatodendritic domain of neurons is mediated by recognition of signals in the cytosolic domains of the proteins by adaptors. These adaptors are components of protein coats associated with the trans-Golgi network and/or recycling endosomes. The clathrin-associated adaptor protein 1 (AP-1) complex plays a preeminent role in this process, although other adaptors and coat proteins, such as AP-4, ARH, Numb, exomer, and retromer, have also been implicated. Cargo adaptor recruitment to the appropriate membrane surface is tightly regulated: in addition to binding cargo, cargo adaptors usually bind directly to the membrane, with membrane specificity enforced through interaction with an organelle-specific phosphoinositide lipid and/or a small GTPase. Cargo adaptors usually also bind to the structural scaffold of a vesicle coat (such as clathrin).
Signal transducing adaptor proteins (STAPs) are accessory to main proteins in a signal transduction pathway. These proteins tend to lack any intrinsic enzymatic activity themselves instead mediating specific protein–protein interactions that drive the formation of protein complexes. Binding of signaling enzymes to such complexes is often dependent on cell stimulation and is mediated by proteins that are collectively termed adaptor proteins or adaptors. Adaptors are usually devoid of catalytic activity, but contain at least two domains that mediate protein–protein interactions. This context-dependent signal transduction is an essential means by which cells handle multiple stimuli. Therefore, elucidating the molecular nature of the interaction of each signaling protein with its adaptor(s) is required for understanding individual signaling pathway and the responses of the cells to both single and multiple stimuli. Examples of adaptor proteins include MYD88, Grb2 and SHC1. Much of the specificity of signal transduction depends on the recruitment of several signaling components such as protein kinases and G-protein GTPases into short-lived active complexes in response to an activating signal such as a growth factor binding to its receptor. TAB2, a novel TAK1- and TRAF6-binding protein that functions as an IL-1 signaling molecule, was showed to translocate from the membrane to the cytosol following IL-1 stimulation and facilitates the interaction between TRAF6 and TAK1. These results suggest that TAB2 functions as an adaptor in the IL-1 signaling pathway to link TAK1 to TRAF6.
Adaptor Proteins in Disease
Many studies suggest that the regulation of both PKC isozymes and their corresponding RACKs is altered in the neoplastic process. Therefore, agents that selectively interfere with or induce these PKC-RACK interactions may provide a new therapeutic for cancer. Because RACKs are also adaptor proteins for several other signaling proteins that can modulate the neoplastic process, regulation of their interaction with RACKs may also provide a useful target for new drug development for the treatment or prevention of cancer.
AP-2/CCVs are involved in autosomal recessive hypercholesterolemia through the associated low-density lipoprotein receptor adapter protein 1. Retromer is involved in recycling components of the plasma membrane. The importance of that recycling at a synapse is hinted at in one of the figures in the gallery. There are at least 3 ways in which retromer dysfunction can contribute to brain disorders, including Alzheimer and Parkinson diseases.
AP-5 is the most recently described complex, and one reason supporting the idea that it is an authentic adaptor complex is that it is associated with hereditary spastic paraplegia, as is AP-4. AP-1 is linked to MEDNIK syndrome. AP-3 is linked to Hermansky–Pudlak syndrome. COPI is linked to an autoimmune disease. COPII is linked to cranio–lenticulo–sutural dysplasia. One of the GGA proteins may be involved in Alzheimer's disease.
The important of adaptor in signal transduction results in the identification of novel therapeutic targets. Over the past years, two parallel strategies have emerged for modifying signaling pathway by targeting adaptor protein–receptor interactions. The first one is targeting the complexes through the use of interfering peptides, which competitively block protein-protein interactions. Recent studies on D2 and 5-HT2C receptors have demonstrated the feasibility of this concept in rodents in vivo. Another approach for this purpose may involve studies of currently used monoaminergic pharmacotherapies and correlate physiological responses with the agonistic or antagonistic properties of these pharmacotherapies on signaling transduction pathways.
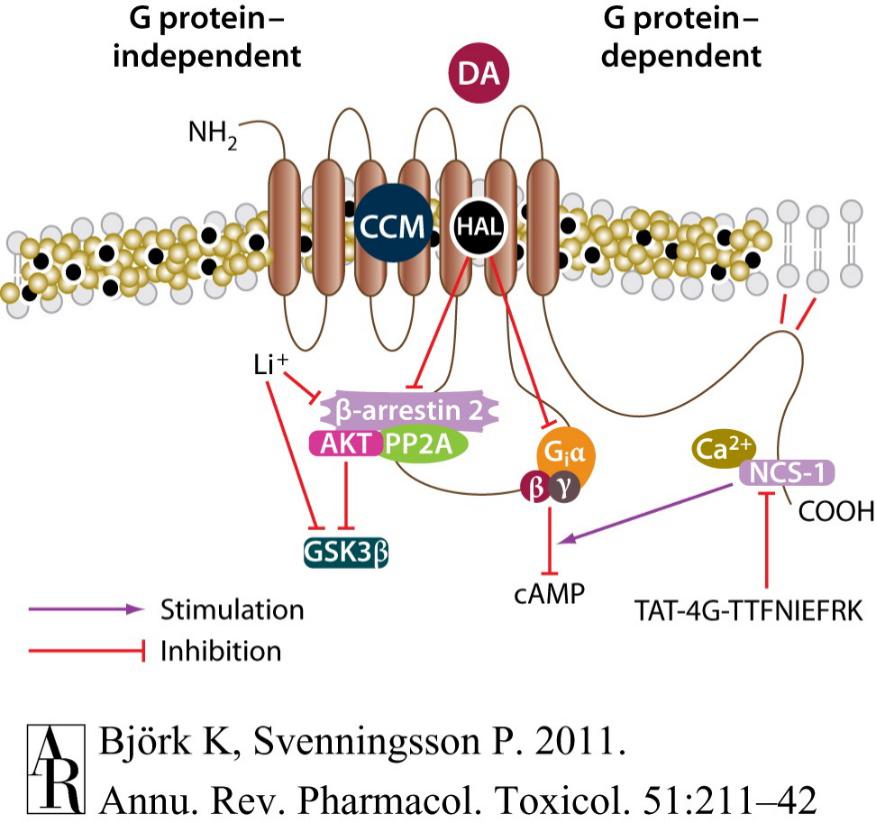
Figure 3. Targeting adaptor proteins and lipid rafts affects response and distribution of pharmacological agents at the D2 receptor
References: