Regulatory status: For research use only, not for use in diagnostic procedures.


| Product Name | Cat. No. | Applications | Host Species | Datasheet | Price | Add to Basket |
|---|
| Product Name | Cat. No. | Applications | Host Species | Datasheet | Price | Add to Basket |
|---|
The discovery of Zonulin arose from the study of Vibrio cholerae. During the development of the Vibrio cholerae vaccine, some researchers found that while the virulence of the initial recombinant Vibrio cholerae vaccine strains that removed the ctxA gene was dramatically reduced, these attenuated strains were still capable of inducing severe diarrhea even in the absence of cholera toxin. To explain this phenomenon, the researchers proposed zonula occludins toxin (Zot), a protein that regulates intestinal tight junction (TJ) exogenous toxins depolymerize the tightly junctioned structures of the intestine, which allows for increased intestinal epithelial permeability. After removal of Zot, the tight junction structure is restored. Thus, Zot is thought to regulate tight junctions in a rapidly reversible manner. The researchers then further used an affinity chromatography column with immobilized Zot antibody to isolate and purify adhesion proteins from human small intestinal tissues, and obtained an endogenous protein that functionally resembles Zot to depolymerize tightly-connected complexes, and named it zonulin.
Zonulin is a protein with a molecular mass of 47kDa, with a specific N-terminal receptor-binding domain that determines tissue specificity, and a C-terminal tau-like domain that correlates with cytoskeletal rearrangements in intercellular TJs. Proteomic analysis identified zonulin as a precursor of haptoglobin 2. This is the only human endogenous protein that has been identified that can regulate intestinal permeability by affecting the tight junction structure of the intestinal epithelium. This protein can depolymerize the intestinal epithelial tight junction structural complexes, leading to disruption of the intercellular tight junction structures and increased permeability of the intestinal epithelium, which can lead to inflammation by allowing the entry of macromolecular antigenic substances into the tissues in the intestine.
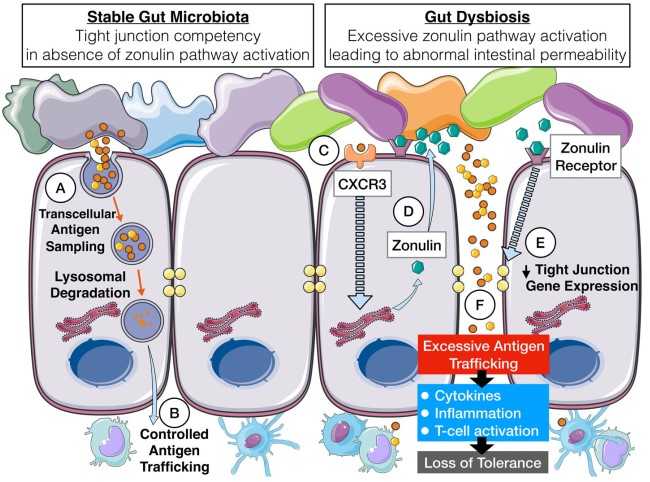 Figure 1. Mechanisms of intestinal dysbiosis due to unregulated zonulin
Figure 1. Mechanisms of intestinal dysbiosis due to unregulated zonulin
(Source: Heickman LK, et al. 2020)
On the other hand, studies have confirmed that zonulin is not only found in the intestinal tract, but also in various tissues and organs such as the heart and brain. However, zonulin extracted from different tissues and organs differed in their N-termini, suggesting that connexins can respond specifically to particular stimuli as needed. Therefore, the researchers hypothesized that zonulin is not only able to regulate the tight junctions between intestinal epithelial cells, but is also involved in regulating the tight junctions between extra-intestinal epithelial cells and vascular endothelial cells.
References
1. Veres-Székely A, et al. Zonulin as a Potential Therapeutic Target in Microbiota-Gut-Brain Axis Disorders: Encouraging Results and Emerging Questions. Int J Mol Sci. 2023 Apr Wood
2. Heickman LK, et al. Zonulin as a potential putative biomarker of risk for shared type 1 diabetes and celiac disease autoimmunity. Diabetes Metab Res Rev. 2020 Jul;36(5):e3309.19;24(8):7548.
Q: Concentration of sodium azide or thimerosal and 2-mercaptoethanol(or other toxic substances) if it is contained in the product (we would appreciate it if you would send us the documentation to confirm the product).
A: According to the attached MSDS for this product, none of the reagents contain Sodium Azide.
Q: DEIA2225 Human Zonulin ELISA Kit was produced with Baculovirus to follow Cartagena protocol regulation (or not).
A: The standard and the immunogens for the antibodies provided in the kit were not expressed with baculo virus.
Q: The protein in the Standard of this product is recombinant (or not).
A: The standard is not a recombinant protein, it is human haptoglobin from Human serum.
Effects of PrObiotics on the Symptoms and Surgical ouTComes after Anterior REsection of Colon Cancer (POSTCARE): A Randomized, Double-Blind, Placebo-Controlled Trial
JOURNAL OF CLINICAL MEDICINE
Authors: Park, In Ja; Lee, Ju-Hoon; Kye, Bong-Hyeon; Oh, Heung-Kwon; Cho, Yong Beom; Kim, You-Tae; Kim, Joo Yun; Sung, Na Young; Kang, Sung-Bum; Seo, Jeong-Meen; Sim, Jae-Hun; Lee, Jung-Lyoul; Lee, In Kyu
IFN-gamma, IL-17A, or zonulin rapidly increase the permeability of the blood-brain and small intestinal epithelial barriers: Relevance for neuro-inflammatory diseases
BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS
Authors: Rahman, Mohammed T.; Ghosh, Chaitali; Hossain, Mohammed; Linfield, Debra; Rezaee, Fariba; Janigro, Damir; Marchi, Nicola; van Boxel-Dezaire, Anette H. H.
![]()