What Is Virus-Like Particles?
Virus-like particles (VLPs) consist of all or part of a protein that forms a viral capsid but lacks a genomic material, similar to the structure of a virus, but cannot replicate in cells, precluding any possibility of reversing mutations or pathogen infection. VLPs are unable to replicate in the receptor, but it stimulates the immune system by recognizing repeating subunits, producing high01 cellular and humoral immune responses. VLPs have antigenicity and immunogenicity similar to natural virions. Therefore, VLP has received a lot of attention as a vaccine candidate because of its advantages over other types of vaccines.
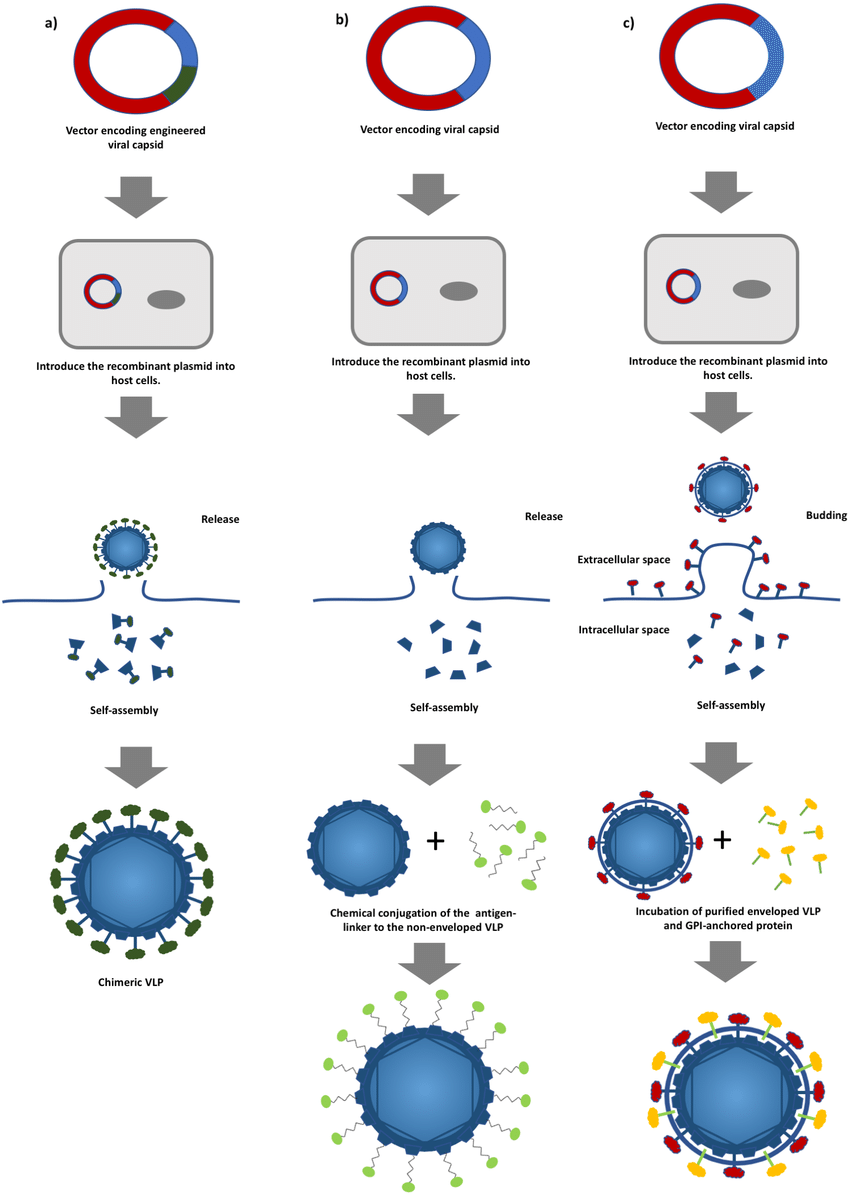
How Are VLPs Produced?
As an artificial synthetic recombinant protein, VLPs need to be synthesized in a specific expression system. A variety of expression systems exist to produce VLPs, including bacterial, yeast, insect and mammalian cells. Since the characteristics of the selected expression system directly affect the characteristics of the produced VLP, one crucial step for successful VLPs production lies in the proper choice of appropriate expression host system. Among them, E. coli is the most widely used classical expression system due to its clear genetic background, rapid propagation, low cost, high expression, easy purification of expression products, good stability, and strong anti-pollution ability.
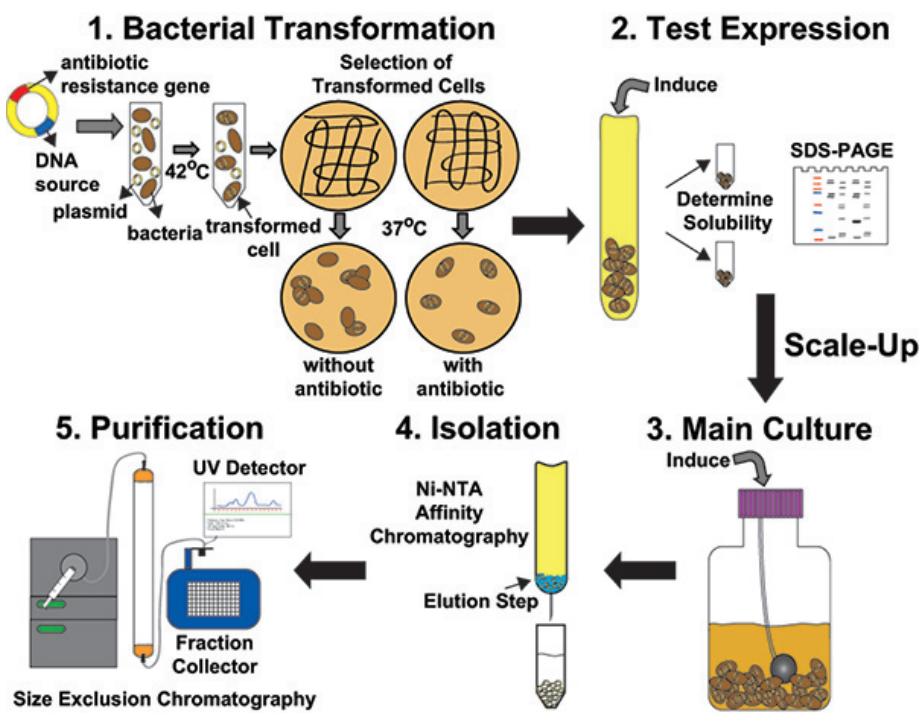
However, recombinant proteins of eukaryotic hosts often appear insoluble and lack proper disulfide bonds in E. coli cells system. Creative Diagnostics has summed up and utilized a series of solutions to conquer these obstacles:
Unable to Produce Proper Disulfide Bonds in Proteins
Due to the catalytic action of the disulfide bond oxidoreductase system, the periplasm is usually the only place in E. coli that can form disulfide bonds. Therefore, if the recombinant protein needs to form a disulfide bond, it needs to be localized to the periplasm using a cleavable signal peptide such as pel B.
Unable to Produce Soluble Proteins in Colicells System
- Lower culture temperature, a very usual way to stimulate the formation of soluble VLPs in colicells.
- Choose the most appropriate expression plasmids with a suitable resistance marker and adjust the composition of the cultivation medium to get a better solubility of recombinant proteins.
- By fusing a tagged protein such as glutathione-S-transferase (GST) to the interested protein to achieve higher expression levels and solubility.
Creative Diagnostics uses the E. coli cell system to create the world’s leading manufacturing platform for virus-like particles (VLPs). The platform can be customized to produce a variety of high purity VLPs. After years of exploration, our scientists have successfully obtained a variety of VLPs for antibody discovery, vaccine development, and diagnostic reagent development. Among them, HBV recombinant VLPs have developed ready-to-use products that can be directly used in your research.
| Catalog# | Product Description | Expression System | Application |
| DAGF-227 | Recombinant HPV type 6 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-228 | Recombinant HPV type 11 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-229 | Recombinant HPV type 16 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-230 | Recombinant HPV type 18 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-231 | Recombinant HPV type 31 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-232 | Recombinant HPV type 33 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-233 | Recombinant HPV type 45 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-234 | Recombinant HPV type 52 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
| DAGF-235 | Recombinant HPV type 58 L1 protein (VLP) | E. Coli | ELISA, Antibody Detection |
